Protease-activated receptor-2 signaling triggers dendritic cell development
- PMID: 12759239
- PMCID: PMC1868121
- DOI: 10.1016/S0002-9440(10)64316-7
Protease-activated receptor-2 signaling triggers dendritic cell development
Abstract
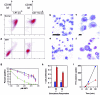
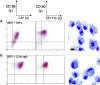
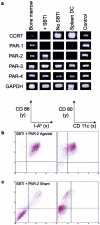
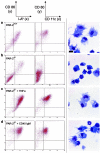
Dendritic cells (DC) are potent antigen-presenting cells that govern the effector cell responses of the immune system. DC are thought to continuously develop from circulating progenitors in a process that is accelerated by inflammatory stimuli. However, the physiological signals that regulate the development of DC from precursor cells have not been well defined. Here we show that a serine protease acting via protease-activated receptor-2 (PAR-2) stimulates the development of DC from bone marrow progenitor cells cultured in granulocyte-macrophage colony-stimulating factor and IL-4. DC fail to develop in bone marrow cultures treated with soy bean trypsin inhibitor, a serine protease inhibitor, but this inhibition is overcome by a PAR-2 agonist peptide. DC do not spontaneously develop from the bone marrow of PAR-2-deficient mice, but can be stimulated to do so by inflammatory mediators. These results suggest that endogenous serine proteases stimulate DC development in vitro. Thus, serine proteases may help trigger adaptive immune responses in vivo.
Figures
References
-
- Steinman RM: The dendritic cell system and its role in immunogenicity. Annu Rev Immunol 1991, 9:271-296 - PubMed
-
- Banchereau J, Steinman RM: Dendritic cells and the control of immunity. Nature 1998, 392:245-252 - PubMed
-
- Cella M, Engering A, Pinet V, Pieters J, Lanzavecchia A: Inflammatory stimuli induce accumulation of MHC class II complexes on dendritic cells. Nature 1997, 388:782-787 - PubMed
-
- Matzinger P: An innate sense of danger. Semin Immunol 1998, 10:399-415 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

