An electronic effect on protein structure
- PMID: 12761389
- PMCID: PMC2323894
- DOI: 10.1110/ps.0241903
An electronic effect on protein structure
Abstract
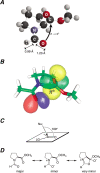
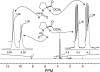
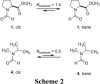
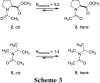
The well-known preference of the peptide bond for the trans conformation has been attributed to steric effects. Here, we show that a proline residue with an N-formyl group (H(i-1)-C'(i-1)=O(i-1)), in which H(i-1) presents less steric hindrance than does O(i-1), likewise prefers a trans conformation. Thus, the preference of the peptide bond for the trans conformation cannot be explained by steric effects alone. Rather, an n --> pi* interaction between the oxygen of the peptide bond (O(i-1)), and the subsequent carbonyl carbon in the polypeptide chain (C'(i)) also contributes to this preference. The O(i-1) and C'(i) distance and O(i-1).C'(i)=O(i) angle are especially favorable for such an n --> pi* interaction in a polyproline II helix. We propose that this electronic effect provides substantial stabilization to this and other elements of protein structure.
Figures


References
-
- An, S.S.A., Lester, C.C., Peng, J.-L., Li, Y.-J., Rothwarf, D.M., Welker, E., Thannhauser, T.W., Zhang, L.S., Tam, J.P., and Scheraga, H.A. 1999. Retention of the cis proline conformation in tripeptide fragments of bovine pancreatic ribonuclease A containing a non-natural proline analogue, 5,5-dimethylproline. J. Am. Chem. Soc. 121 11558–11566.
-
- Antonyraj, K.J., Karunakaran, T., and Raj, P.A. 1998. Bactericidal activity and poly-L-proline II conformation of the tandem repeat sequence of human salivary mucin glycoprotein (MG2). Arch. Biochem. Biophys. 356 197–206. - PubMed
-
- Baldwin, J.E. 1976. Approach vector analysis: A stereochemical approach to reactivity. Chem. Commun. 738–741.
-
- Bella, J., Eaton, M., Brodsky, B., and Berman, H.M. 1994. Crystal and molecular structure of a collagen-like peptide at 1.9 Å resolution. Science 266 75–81. - PubMed
-
- Benzi, C., Improta, R., Scalmani, G., and Barone, V. 2002. Quantum mechanical study of the conformational behavior of proline and 4R-hydroxyproline dipeptide analogues in vacuum and in aqueous solution. J. Comput. Chem. 23 341–350. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

