Protein-protein docking with a reduced protein model accounting for side-chain flexibility
- PMID: 12761398
- PMCID: PMC2323887
- DOI: 10.1110/ps.0239303
Protein-protein docking with a reduced protein model accounting for side-chain flexibility
Abstract
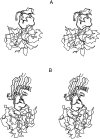
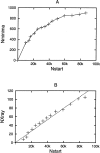
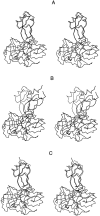
A protein-protein docking approach has been developed based on a reduced protein representation with up to three pseudo atoms per amino acid residue. Docking is performed by energy minimization in rotational and translational degrees of freedom. The reduced protein representation allows an efficient search for docking minima on the protein surfaces within. During docking, an effective energy function between pseudo atoms has been used based on amino acid size and physico-chemical character. Energy minimization of protein test complexes in the reduced representation results in geometries close to experiment with backbone root mean square deviations (RMSDs) of approximately 1 to 3 A for the mobile protein partner from the experimental geometry. For most test cases, the energy-minimized experimental structure scores among the top five energy minima in systematic docking studies when using both partners in their bound conformations. To account for side-chain conformational changes in case of using unbound protein conformations, a multicopy approach has been used to select the most favorable side-chain conformation during the docking process. The multicopy approach significantly improves the docking performance, using unbound (apo) binding partners without a significant increase in computer time. For most docking test systems using unbound partners, and without accounting for any information about the known binding geometry, a solution within approximately 2 to 3.5 A RMSD of the full mobile partner from the experimental geometry was found among the 40 top-scoring complexes. The approach could be extended to include protein loop flexibility, and might also be useful for docking of modeled protein structures.
Figures
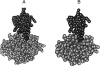
Similar articles
-
Progress in protein-protein docking: atomic resolution predictions in the CAPRI experiment using RosettaDock with an improved treatment of side-chain flexibility.Proteins. 2005 Aug 1;60(2):187-94. doi: 10.1002/prot.20556. Proteins. 2005. PMID: 15981249
-
Rapid refinement of protein interfaces incorporating solvation: application to the docking problem.J Mol Biol. 1998 Feb 13;276(1):265-85. doi: 10.1006/jmbi.1997.1519. J Mol Biol. 1998. PMID: 9514726
-
ATTRACT: protein-protein docking in CAPRI using a reduced protein model.Proteins. 2005 Aug 1;60(2):252-6. doi: 10.1002/prot.20566. Proteins. 2005. PMID: 15981270
-
Recognition-induced conformational changes in protein-protein docking.Curr Pharm Biotechnol. 2008 Apr;9(2):77-86. doi: 10.2174/138920108783955173. Curr Pharm Biotechnol. 2008. PMID: 18393864 Review.
-
Flexible protein-protein docking.Curr Opin Struct Biol. 2006 Apr;16(2):194-200. doi: 10.1016/j.sbi.2006.02.002. Epub 2006 Feb 17. Curr Opin Struct Biol. 2006. PMID: 16488145 Review.
Cited by
-
Exploring Conformational Landscapes and Cryptic Binding Pockets in Distinct Functional States of the SARS-CoV-2 Omicron BA.1 and BA.2 Trimers: Mutation-Induced Modulation of Protein Dynamics and Network-Guided Prediction of Variant-Specific Allosteric Binding Sites.Viruses. 2023 Sep 27;15(10):2009. doi: 10.3390/v15102009. Viruses. 2023. PMID: 37896786 Free PMC article.
-
PTools: an opensource molecular docking library.BMC Struct Biol. 2009 May 1;9:27. doi: 10.1186/1472-6807-9-27. BMC Struct Biol. 2009. PMID: 19409097 Free PMC article.
-
Accounting for large amplitude protein deformation during in silico macromolecular docking.Int J Mol Sci. 2011 Feb 22;12(2):1316-33. doi: 10.3390/ijms12021316. Int J Mol Sci. 2011. PMID: 21541061 Free PMC article.
-
Integrative modeling of membrane-associated protein assemblies.Nat Commun. 2020 Dec 4;11(1):6210. doi: 10.1038/s41467-020-20076-5. Nat Commun. 2020. PMID: 33277503 Free PMC article.
-
A Database of Force-Field Parameters, Dynamics, and Properties of Antimicrobial Compounds.Molecules. 2015 Aug 3;20(8):13997-4021. doi: 10.3390/molecules200813997. Molecules. 2015. PMID: 26247924 Free PMC article.
References
-
- Ausiello, G., Cesareni, G., and Helmer-Citterich, M. 1997. A new docking procedure applied to reconstruction of protein tertiary structure. Proteins 28 556–567. - PubMed
-
- Bernstein, F.C., Koetzle, T.F., Williams, G.J., Meyer, E.E., Brice, M.D., Rodgers, J.R., Kennard, O., Shianouchi, T., and Tasumi, M. 1977. The PDB: A computer-based archival file for macromolecular structures. J. Mol. Biol. 112 535–542. - PubMed
-
- Betts, M.J. and Sternberg, M.J.E. 1999. An analysis of conformational changes on protein–protein association: Implications for predictive docking. Protein Eng. 12 271–283. - PubMed
-
- Camacho, J.C., Gatchell, D.W., Kimura, S.R., and Vajda, S. 2000. Scoring docked conformations generated by rigid-body protein–protein docking. Proteins 40 525–537. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

