Molecular dynamics simulation of a dipalmitoylphosphatidylcholine bilayer with NaCl
- PMID: 12770880
- PMCID: PMC1302956
- DOI: 10.1016/S0006-3495(03)75102-9
Molecular dynamics simulation of a dipalmitoylphosphatidylcholine bilayer with NaCl
Abstract
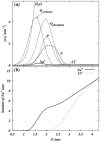
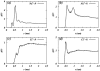
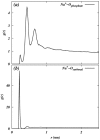
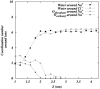
Molecular dynamics simulations are performed on two hydrated dipalmitoylphosphatidylcholine bilayer systems: one with pure water and one with added NaCl. Due to the rugged nature of the membrane/electrolyte interface, ion binding to the membrane surface is characterized by the loss of ion hydration. Using this structural characterization, binding of Na(+) and Cl(-) ions to the membrane is observed, although the binding of Cl(-) is seen to be slightly weaker than that of Na(+). Dehydration is seen to occur to a different extent for each type of ion. In addition, the excess binding of Na(+) gives rise to a net positive surface charge density just outside the bilayer. The positive density produces a positive electrostatic potential in this region, whereas the system without salt shows an electrostatic potential of zero.
Figures
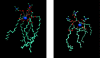

Similar articles
-
Mixed bilayer containing dipalmitoylphosphatidylcholine and dipalmitoylphosphatidylserine: lipid complexation, ion binding, and electrostatics.Biophys J. 2003 Nov;85(5):3120-31. doi: 10.1016/S0006-3495(03)74730-4. Biophys J. 2003. PMID: 14581212 Free PMC article.
-
Molecular dynamics simulation of a palmitoyl-oleoyl phosphatidylserine bilayer with Na+ counterions and NaCl.Biophys J. 2004 Mar;86(3):1601-9. doi: 10.1016/S0006-3495(04)74227-7. Biophys J. 2004. PMID: 14990486 Free PMC article.
-
Study of the effect of Na+ and Ca2+ ion concentration on the structure of an asymmetric DPPC/DPPC + DPPS lipid bilayer by molecular dynamics simulation.Colloids Surf B Biointerfaces. 2009 Oct 1;73(1):42-50. doi: 10.1016/j.colsurfb.2009.04.028. Epub 2009 May 9. Colloids Surf B Biointerfaces. 2009. PMID: 19487110
-
Ion dynamics in cationic lipid bilayer systems in saline solutions.J Phys Chem B. 2009 Jul 9;113(27):9226-34. doi: 10.1021/jp810233q. J Phys Chem B. 2009. PMID: 19534449
-
A computer perspective of membranes: molecular dynamics studies of lipid bilayer systems.Biochim Biophys Acta. 1997 Nov 21;1331(3):235-70. doi: 10.1016/s0304-4157(97)00008-7. Biochim Biophys Acta. 1997. PMID: 9512654 Review. No abstract available.
Cited by
-
Protein surface dynamics: interaction with water and small solutes.J Biol Phys. 2005 Dec;31(3-4):433-52. doi: 10.1007/s10867-005-0171-2. J Biol Phys. 2005. PMID: 23345909 Free PMC article.
-
How alcohol chain-length and concentration modulate hydrogen bond formation in a lipid bilayer.Biophys J. 2007 Apr 1;92(7):2366-76. doi: 10.1529/biophysj.106.097022. Epub 2007 Jan 11. Biophys J. 2007. PMID: 17218462 Free PMC article.
-
Molecular dynamics simulations of a DMPC bilayer using nonadditive interaction models.Biophys J. 2009 Jan;96(2):385-402. doi: 10.1016/j.bpj.2008.09.048. Biophys J. 2009. PMID: 19167291 Free PMC article.
-
Test of the Gouy-Chapman theory for a charged lipid membrane against explicit-solvent molecular dynamics simulations.Phys Rev Lett. 2008 Jul 18;101(3):038103. doi: 10.1103/PhysRevLett.101.038103. Epub 2008 Jul 18. Phys Rev Lett. 2008. PMID: 18764300 Free PMC article.
-
Toward the discovery of vaccine adjuvants: coupling in silico screening and in vitro analysis of antagonist binding to human and mouse CCR4 receptors.PLoS One. 2009 Nov 30;4(11):e8084. doi: 10.1371/journal.pone.0008084. PLoS One. 2009. PMID: 20011659 Free PMC article.
References
-
- Berendsen, H., D. van der Spoel, and R. van Drunen. 1995. GROMACS: a message-passing parallel molecular dynamics implementation. Comp. Phys. Comm. 91:43–56.
-
- Berg, O. G., J. Rogers, B-Z. Yu, J. Yao, L. S. Romsted, and M. K. Jain. 1997. Thermodynamic and kinetic basis of interfacial activation: resolution of binding and allosteric effects on pancreatic phospholipase A2 at zwitterionic interfaces. Biochem. 36:14512–14530. - PubMed
-
- Binder, H., and O. Zschörnig. 2002. The effect of metal cations on the phase behavior and hydration characteristics of phospholipid membranes. Chem. Phys. Lipids. 115:39–61. - PubMed
-
- Cevc, G. 1990. Membrane electrostatics. Biochim. Biophys. Acta. 1031:311–382. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

