Saxitoxin is a gating modifier of HERG K+ channels
- PMID: 12771193
- PMCID: PMC2217357
- DOI: 10.1085/jgp.200308812
Saxitoxin is a gating modifier of HERG K+ channels
Abstract
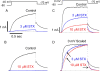
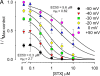
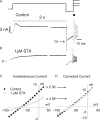
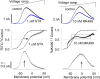
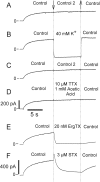
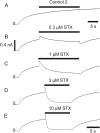
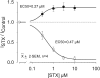
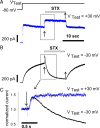
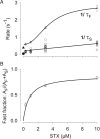
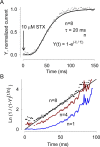
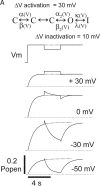
Potassium (K+) channels mediate numerous electrical events in excitable cells, including cellular membrane potential repolarization. The hERG K+ channel plays an important role in myocardial repolarization, and inhibition of these K+ channels is associated with long QT syndromes that can cause fatal cardiac arrhythmias. In this study, we identify saxitoxin (STX) as a hERG channel modifier and investigate the mechanism using heterologous expression of the recombinant channel in HEK293 cells. In the presence of STX, channels opened slower during strong depolarizations, and they closed much faster upon repolarization, suggesting that toxin-bound channels can still open but are modified, and that STX does not simply block the ion conduction pore. STX decreased hERG K+ currents by stabilizing closed channel states visualized as shifts in the voltage dependence of channel opening to more depolarized membrane potentials. The concentration dependence for steady-state modification as well as the kinetics of onset and recovery indicate that multiple STX molecules bind to the channel. Rapid application of STX revealed an apparent "agonist-like" effect in which K+ currents were transiently increased. The mechanism of this effect was found to be an effect on the channel voltage-inactivation relationship. Because the kinetics of inactivation are rapid relative to activation for this channel, the increase in K+ current appeared quickly and could be subverted by a decrease in K+ currents due to the shift in the voltage-activation relationship at some membrane potentials. The results are consistent with a simple model in which STX binds to the hERG K+ channel at multiple sites and alters the energetics of channel gating by shifting both the voltage-inactivation and voltage-activation processes. The results suggest a novel extracellular mechanism for pharmacological manipulation of this channel through allosteric coupling to channel gating.
Figures






Similar articles
-
Altered gating of HERG potassium channels by cobalt and lanthanum.Pflugers Arch. 2000 Jun;440(2):264-74. doi: 10.1007/s004240000263. Pflugers Arch. 2000. PMID: 10898527
-
Interactions of the antimalarial drug mefloquine with the human cardiac potassium channels KvLQT1/minK and HERG.J Pharmacol Exp Ther. 2001 Oct;299(1):290-6. J Pharmacol Exp Ther. 2001. PMID: 11561091
-
BeKm-1 is a HERG-specific toxin that shares the structure with ChTx but the mechanism of action with ErgTx1.Biophys J. 2003 May;84(5):3022-36. doi: 10.1016/S0006-3495(03)70028-9. Biophys J. 2003. PMID: 12719233 Free PMC article.
-
Biophysical properties and molecular basis of cardiac rapid and slow delayed rectifier potassium channels.Cell Physiol Biochem. 1999;9(4-5):201-16. doi: 10.1159/000016317. Cell Physiol Biochem. 1999. PMID: 10575198 Review.
-
Familial and acquired long qt syndrome and the cardiac rapid delayed rectifier potassium current.Clin Exp Pharmacol Physiol. 2000 Oct;27(10):753-66. doi: 10.1046/j.1440-1681.2000.03337.x. Clin Exp Pharmacol Physiol. 2000. PMID: 11022966 Review.
Cited by
-
Behavioral alterations induced by repeated saxitoxin exposure in drinking water.J Venom Anim Toxins Incl Trop Dis. 2016 May 17;22:18. doi: 10.1186/s40409-016-0072-9. eCollection 2016. J Venom Anim Toxins Incl Trop Dis. 2016. PMID: 27190499 Free PMC article.
-
The Genetic Basis of Toxin Biosynthesis in Dinoflagellates.Microorganisms. 2019 Jul 29;7(8):222. doi: 10.3390/microorganisms7080222. Microorganisms. 2019. PMID: 31362398 Free PMC article. Review.
-
Current Trends and New Challenges in Marine Phycotoxins.Mar Drugs. 2022 Mar 8;20(3):198. doi: 10.3390/md20030198. Mar Drugs. 2022. PMID: 35323497 Free PMC article. Review.
-
Structure of the saxiphilin:saxitoxin (STX) complex reveals a convergent molecular recognition strategy for paralytic toxins.Sci Adv. 2019 Jun 19;5(6):eaax2650. doi: 10.1126/sciadv.aax2650. eCollection 2019 Jun. Sci Adv. 2019. PMID: 31223657 Free PMC article.
-
Alexandrium spp.: From Toxicity to Potential Biotechnological Benefits.Mar Drugs. 2023 Dec 30;22(1):31. doi: 10.3390/md22010031. Mar Drugs. 2023. PMID: 38248656 Free PMC article. Review.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

