In vivo two-photon calcium imaging of neuronal networks
- PMID: 12777621
- PMCID: PMC165873
- DOI: 10.1073/pnas.1232232100
In vivo two-photon calcium imaging of neuronal networks
Abstract
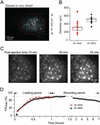
Two-photon calcium imaging is a powerful means for monitoring the activity of distinct neurons in brain tissue in vivo. In the mammalian brain, such imaging studies have been restricted largely to calcium recordings from neurons that were individually dye-loaded through microelectrodes. Previous attempts to use membrane-permeant forms of fluorometric calcium indicators to load populations of neurons have yielded satisfactory results only in cell cultures or in slices of immature brain tissue. Here we introduce a versatile approach for loading membrane-permeant fluorescent indicator dyes in large populations of cells. We established a pressure ejection-based local dye delivery protocol that can be used for a large spectrum of membrane-permeant indicator dyes, including calcium green-1 acetoxymethyl (AM) ester, Fura-2 AM, Fluo-4 AM, and Indo-1 AM. We applied this dye-loading protocol successfully in mouse brain tissue at any developmental stage from newborn to adult in vivo and in vitro. In vivo two-photon Ca2+ recordings, obtained by imaging through the intact skull, indicated that whisker deflection-evoked Ca2+ transients occur in a subset of layer 2/3 neurons of the barrel cortex. Thus, our results demonstrate the suitability of this technique for real-time analyses of intact neuronal circuits with the resolution of individual cells.
Figures




References
-
- Nicolelis, M. & Ribeiro, S. (2002) Curr. Opin. Neurobiol. 12, 602-606. - PubMed
-
- Shoham, D., Glaser, D. E., Arieli, A., Kenet, T., Wijnbergen, C., Toledo, Y., Hildesheim, R. & Grinvald, A. (1999) Neuron 24, 791-802. - PubMed
-
- Grinvald, A., Frostig, R. D., Lieke, E. & Hildesheim, R. (1988) Physiol. Rev. 68, 1285-1366. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

