In vivo administration of a lentiviral vaccine targets DCs and induces efficient CD8(+) T cell responses
- PMID: 12782670
- PMCID: PMC156105
- DOI: 10.1172/JCI17098
In vivo administration of a lentiviral vaccine targets DCs and induces efficient CD8(+) T cell responses
Abstract
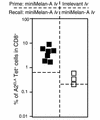
The present study evaluates the potential of third-generation lentivirus vectors with respect to their use as in vivo-administered T cell vaccines. We demonstrate that lentivector injection into the footpad of mice transduces DCs that appear in the draining lymph node and in the spleen. In addition, a lentivector vaccine bearing a T cell antigen induced very strong systemic antigen-specific cytotoxic T lymphocyte (CTL) responses in mice. Comparative vaccination performed in two different antigen models demonstrated that in vivo administration of lentivector was superior to transfer of transduced DCs or peptide/adjuvant vaccination in terms of both amplitude and longevity of the CTL response. Our data suggest that a decisive factor for efficient T cell priming by lentivector might be the targeting of DCs in situ and their subsequent migration to secondary lymphoid organs. The combination of performance, ease of application, and absence of pre-existing immunity in humans make lentivector-based vaccines an attractive candidate for cancer immunotherapy.
Figures



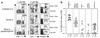

References
-
- Nestle FO, et al. Vaccination of melanoma patients with peptide- or tumor lysate–pulsed dendritic cells. Nat. Med. 1998;4:328–332. - PubMed
-
- Brossart P, Goldrath AW, Butz EA, Martin S, Bevan MJ. Virus-mediated delivery of antigenic epitopes into dendritic cells as a means to induce CTL. J. Immunol. 1997;158:3270–3276. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

