Nanometer targeting of microtubules to focal adhesions
- PMID: 12782685
- PMCID: PMC2172972
- DOI: 10.1083/jcb.200301102
Nanometer targeting of microtubules to focal adhesions
Abstract
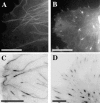
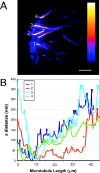
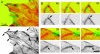
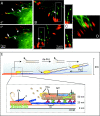
Although cell movement is driven by actin, polarization and directional locomotion require an intact microtubule cytoskeleton that influences polarization by modulating substrate adhesion via specific targeting interactions with adhesion complexes. The fidelity of adhesion site targeting is precise; using total internal reflection fluorescence microscopy (TIRFM), we now show microtubule ends (visualized by incorporation of GFP tubulin) are within 50 nm of the substrate when polymerizing toward the cell periphery, but not when shrinking from it. Multiple microtubules sometimes followed similar tracks, suggesting guidance along a common cytoskeletal element. Use of TIRFM with GFP- or DsRed-zyxin in combination with either GFP-tubulin or GFP-CLIP-170 further revealed that the polymerizing microtubule plus ends that tracked close to the dorsal surface consistently targeted substrate adhesion complexes. This supports a central role for the microtubule tip complex in the guidance of microtubules into adhesion foci, and provides evidence for an intimate cross-talk between microtubule tips and substrate adhesions in the range of molecular dimensions.
Figures

Similar articles
-
Microtubule-targeting-dependent reorganization of filopodia.J Cell Sci. 2007 Apr 1;120(Pt 7):1235-44. doi: 10.1242/jcs.003913. Epub 2007 Mar 13. J Cell Sci. 2007. PMID: 17356063
-
Differential localisation of GFP fusions to cytoskeleton-binding proteins in animal, plant, and yeast cells. Green-fluorescent protein.Protoplasma. 2002 Oct;220(1-2):69-78. doi: 10.1007/s00709-002-0026-7. Protoplasma. 2002. PMID: 12417938
-
The conformational state of Tes regulates its zyxin-dependent recruitment to focal adhesions.J Cell Biol. 2003 Apr 14;161(1):33-9. doi: 10.1083/jcb.200211015. J Cell Biol. 2003. PMID: 12695497 Free PMC article.
-
Microtubule involvement in regulating cell contractility and adhesion-dependent signalling: a possible mechanism for polarization of cell motility.Biochem Soc Symp. 1999;65:147-72. Biochem Soc Symp. 1999. PMID: 10320938 Review.
-
Touch, grasp, deliver and control: functional cross-talk between microtubules and cell adhesions.Traffic. 2009 Mar;10(3):268-74. doi: 10.1111/j.1600-0854.2008.00869.x. Epub 2009 Jan 17. Traffic. 2009. PMID: 19175539 Review.
Cited by
-
The Cytoskeleton-A Complex Interacting Meshwork.Cells. 2019 Apr 18;8(4):362. doi: 10.3390/cells8040362. Cells. 2019. PMID: 31003495 Free PMC article. Review.
-
Profiling distinct mechanisms of tumour invasion for drug discovery: imaging adhesion, signalling and matrix turnover.Clin Exp Metastasis. 2009;26(4):381-97. doi: 10.1007/s10585-008-9222-y. Epub 2008 Oct 29. Clin Exp Metastasis. 2009. PMID: 18958576 Review.
-
Profilin connects actin assembly with microtubule dynamics.Mol Biol Cell. 2016 Aug 1;27(15):2381-93. doi: 10.1091/mbc.E15-11-0799. Epub 2016 Jun 15. Mol Biol Cell. 2016. PMID: 27307590 Free PMC article.
-
APC is a component of an organizing template for cortical microtubule networks.Nat Cell Biol. 2005 May;7(5):463-73. doi: 10.1038/ncb1248. Nat Cell Biol. 2005. PMID: 15892196 Free PMC article.
-
Organization, dynamics and mechanoregulation of integrin-mediated cell-ECM adhesions.Nat Rev Mol Cell Biol. 2023 Feb;24(2):142-161. doi: 10.1038/s41580-022-00531-5. Epub 2022 Sep 27. Nat Rev Mol Cell Biol. 2023. PMID: 36168065 Free PMC article. Review.
References
-
- Axelrod, D. 1989. Total internal reflection fluorescence microscopy. Methods Cell Biol. 30:245–270. - PubMed
-
- Gavin, R.H. 1997. Microtubule-microfilament synergy in the cytoskeleton. Int. Rev. Cytol. 173:207–242. - PubMed
-
- Geiger, B., and A. Bershadsky. 2001. Assembly and mechanosensory function of focal contacts. Curr. Opin. Cell Biol. 13:584–592. - PubMed
-
- Goode, B.L., D.G. Drubin, and G. Barnes. 2000. Functional cooperation between the microtubule and actin cytoskeletons. Curr. Opin. Cell Biol. 12:63–71. - PubMed
-
- Gundersen, G.G. 2002. Evolutionary conservation of microtubule-capture mechanisms. Nat. Rev. Mol. Cell Biol. 3:296–304. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

