PG-mediated closure of paracellular pathway and not restitution is the primary determinant of barrier recovery in acutely injured porcine ileum
- PMID: 12801887
- PMCID: PMC2443786
- DOI: 10.1152/ajpgi.00532.2002
PG-mediated closure of paracellular pathway and not restitution is the primary determinant of barrier recovery in acutely injured porcine ileum
Abstract
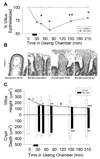
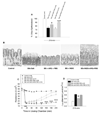
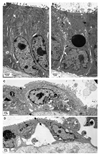
Small bowel epithelium is at the frontline of intestinal barrier function. Restitution is considered to be the major determinant of epithelial repair, because function recovers in parallel with restitution after acute injury. As such, studies of intact mucosa have largely been replaced by migration assays of cultured epithelia. These latter studies fail to account for the simultaneous roles played by villous contraction and paracellular permeability in recovery of barrier function. NSAIDs result in increased intestinal permeability and disease exacerbation in patients with inflammatory bowel disease (IBD). Thus we examined the reparative attributes of endogenous PGs after injury of ileal mucosa by deoxycholate (6 mM) in Ussing chambers. Recovery of transepithelial electrical resistance (TER) from 20-40 Omega.cm2 was abolished by indomethacin (Indo), whereas restitution of 40-100% of the villous surface was unaffected despite concurrent arrest of villous contraction. In the presence of PG, resident crypt and migrating epithelial cells were tightly apposed. In tissues treated with Indo, crypt epithelial cells had dilated intercellular spaces that were accentuated in the migrating epithelium. TER was fully rescued from the effects of Indo by osmotic-driven collapse of the paracellular space, and PG-mediated recovery was significantly impaired by blockade of Cl- secretion. These studies are the first to clearly distinguish the relative contribution of paracellular resistance vs. restitution to acute recovery of epithelial barrier function. Restitution was ineffective in the absence of PG-mediated paracellular space closure. Failure of PG-mediated repair mechanisms may underlie barrier failure resulting from NSAID use in patients with underlying enteropathy.
Figures






Similar articles
-
Prostaglandins I2 and E2 have a synergistic role in rescuing epithelial barrier function in porcine ileum.J Clin Invest. 1997 Oct 15;100(8):1928-33. doi: 10.1172/JCI119723. J Clin Invest. 1997. PMID: 9329955 Free PMC article.
-
Physiological concentrations of bile salts inhibit recovery of ischemic-injured porcine ileum.Am J Physiol Gastrointest Liver Physiol. 2004 Aug;287(2):G399-407. doi: 10.1152/ajpgi.00310.2003. Epub 2004 Apr 15. Am J Physiol Gastrointest Liver Physiol. 2004. PMID: 15087278
-
Recovery of mucosal barrier function in ischemic porcine ileum and colon is stimulated by a novel agonist of the ClC-2 chloride channel, lubiprostone.Am J Physiol Gastrointest Liver Physiol. 2007 Feb;292(2):G647-56. doi: 10.1152/ajpgi.00183.2006. Epub 2006 Oct 19. Am J Physiol Gastrointest Liver Physiol. 2007. PMID: 17053162
-
Restoration of barrier function in injured intestinal mucosa.Physiol Rev. 2007 Apr;87(2):545-64. doi: 10.1152/physrev.00012.2006. Physiol Rev. 2007. PMID: 17429041 Review.
-
Epithelial restitution and wound healing in inflammatory bowel disease.World J Gastroenterol. 2008 Jan 21;14(3):348-53. doi: 10.3748/wjg.14.348. World J Gastroenterol. 2008. PMID: 18200658 Free PMC article. Review.
Cited by
-
Epithelial restitution defect in neonatal jejunum is rescued by juvenile mucosal homogenate in a pig model of intestinal ischemic injury and repair.PLoS One. 2018 Aug 23;13(8):e0200674. doi: 10.1371/journal.pone.0200674. eCollection 2018. PLoS One. 2018. PMID: 30138372 Free PMC article.
-
A guide to Ussing chamber studies of mouse intestine.Am J Physiol Gastrointest Liver Physiol. 2009 Jun;296(6):G1151-66. doi: 10.1152/ajpgi.90649.2008. Epub 2009 Apr 2. Am J Physiol Gastrointest Liver Physiol. 2009. PMID: 19342508 Free PMC article. Review.
-
PTGER4 expression-modulating polymorphisms in the 5p13.1 region predispose to Crohn's disease and affect NF-κB and XBP1 binding sites.PLoS One. 2012;7(12):e52873. doi: 10.1371/journal.pone.0052873. Epub 2012 Dec 27. PLoS One. 2012. PMID: 23300802 Free PMC article.
-
The Integral Role of Tight Junction Proteins in the Repair of Injured Intestinal Epithelium.Int J Mol Sci. 2020 Feb 1;21(3):972. doi: 10.3390/ijms21030972. Int J Mol Sci. 2020. PMID: 32024112 Free PMC article. Review.
-
Doxorubicin increases permeability of murine small intestinal epithelium and cultured T84 monolayers.Sci Rep. 2020 Dec 8;10(1):21486. doi: 10.1038/s41598-020-78473-1. Sci Rep. 2020. PMID: 33293626 Free PMC article.
References
-
- Argenzio RA, Henderson CK, Liacos JA. Restitution of barrier and transport function of porcine colon after acute mucosal injury. American Journal of Physiology. 1988;255:G62–G71. - PubMed
-
- Argenzio RA, Liacos JA, Levy ML, Meuten DJ, Lecce JG, Powell DW. Villous atrophy, crypt hyperplasia, cellular infiltration, and impaired glucose-Na absorption in enteric cryptosporidiosis of pigs. Gastroenterology. 1990;98:1129–1140. - PubMed
-
- Bjarnason I, Hayllar J, MacPherson AJ, Russell AS. Side effects of nonsteroidal anti-inflammatory drugs on the small and large intestine in humans. Gastroenterology. 1993;104:1832–1847. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

