Survivin is required for a sustained spindle checkpoint arrest in response to lack of tension
- PMID: 12805209
- PMCID: PMC162159
- DOI: 10.1093/emboj/cdg307
Survivin is required for a sustained spindle checkpoint arrest in response to lack of tension
Abstract
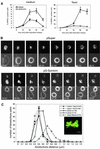
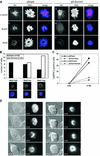
Genetic evidence is mounting that survivin plays a crucial role in mitosis, but its exact role in human cell division remains elusive. We show that mammalian cells lacking survivin are unable to align their chromosomes, fail to recruit Aurora B to kinetochores and become polyploid at a very high frequency. Survivin-depleted cells enter mitosis with normal kinetics, but are delayed in prometaphase in a BubR1/Mad2-dependent fashion. Nonetheless, these cells exit mitosis prior to completion of chromosome congression and without sister chromatid segregation, indicating that the spindle assembly checkpoint is not fully functional. Indeed, in survivin-depleted cells, BubR1 and Mad2 are prematurely displaced from kinetochores, yet no tension is generated at kinetochores. Importantly, these cells fail to respond to drugs that prevent tension, but do arrest in mitosis after depolymerization of the mitotic spindle. This demonstrates that survivin is not required for initial checkpoint activation, or for sustained checkpoint activation by loss of microtubules. However, stable association of BubR1 to kinetochores and sustained checkpoint signalling in response to lack of tension crucially depend on survivin.
Figures








References
-
- Adams R.R., Wheatley,S.P., Gouldsworthy,A.M., Kandels-Lewis,S.E., Carmena,M., Smythe,C., Gerloff,D.L. and Earnshaw,W.C. (2000) INCENP binds the Aurora-related kinase AIRK2 and is required to target it to chromosomes, the central spindle and cleavage furrow. Curr. Biol., 10, 1075–1078. - PubMed
-
- Adams R.R., Carmena,M. and Earnshaw,W.C. (2001) Chromosomal passengers and the (aurora) ABCs of mitosis. Trends Cell Biol., 11, 49–54. - PubMed
-
- Brummelkamp T.R., Bernards,R. and Agami,R. (2002) A system for stable expression of short interfering RNAs in mammalian cells. Science, 296, 550–553. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

