On the mechanism of chaperone activity of the small heat-shock protein of Methanococcus jannaschii
- PMID: 12817080
- PMCID: PMC166198
- DOI: 10.1073/pnas.1032940100
On the mechanism of chaperone activity of the small heat-shock protein of Methanococcus jannaschii
Abstract
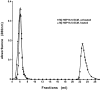
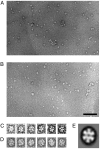
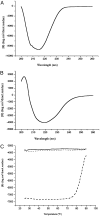
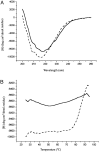
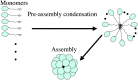
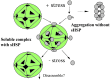
The small heat-shock protein (sHSP) from Methanococcus jannaschii (Mj HSP16.5) forms a homomeric complex of 24 subunits and has an overall structure of a multiwindowed hollow sphere with an external diameter of approximately 120 A and an internal diameter of approximately 65 A with six square "windows" of approximately 17 A across and eight triangular windows of approximately 30 A across. This sHSP has been known to protect other proteins from thermal denaturation. Using purified single-chain monellin as a substrate and a series of methods such as protease digestion, antibody binding, and electron microscopy, we show that the substrates bind to Mj HSP16.5 at a high temperature (80 degrees C) on the outside surface of the sphere and are prevented from forming insoluble substrate aggregates in vitro. Circular dichroism studies suggest that a very small, if any, conformational change occurs in sHSP even at 80 degrees C, but substantial conformational changes of the substrate are required for complex formation at 80 degrees C. Furthermore, deletion mutation studies of Mj HSP16.5 suggest that the N-terminal region of the protein has no structural role but may play an important kinetic role in the assembly of the sphere by "preassembly condensation" of multiple monomers before final assembly of the sphere.
Figures


Similar articles
-
Activation mechanism of HSP16.5 from Methanococcus jannaschii.Biochem Biophys Res Commun. 2003 Aug 8;307(4):991-8. doi: 10.1016/s0006-291x(03)01302-0. Biochem Biophys Res Commun. 2003. PMID: 12878210
-
Small heat shock protein of Methanococcus jannaschii, a hyperthermophile.Proc Natl Acad Sci U S A. 1998 Aug 4;95(16):9129-33. doi: 10.1073/pnas.95.16.9129. Proc Natl Acad Sci U S A. 1998. PMID: 9689045 Free PMC article.
-
Preheating induced homogeneity of the small heat shock protein from Methanococcus jannaschii.Biochim Biophys Acta. 2008 Mar;1784(3):489-95. doi: 10.1016/j.bbapap.2007.12.008. Epub 2008 Jan 3. Biochim Biophys Acta. 2008. PMID: 18211832
-
The minimal α-crystallin domain of Mj Hsp16.5 is functional at non-heat-shock conditions.Proteins. 2014 Jul;82(7):1156-67. doi: 10.1002/prot.24480. Epub 2013 Dec 6. Proteins. 2014. PMID: 24243469
-
Thermal adaptation of heat shock proteins.Curr Protein Pept Sci. 2008 Dec;9(6):552-66. doi: 10.2174/138920308786733903. Curr Protein Pept Sci. 2008. PMID: 19075746 Review.
Cited by
-
Small heat shock proteins: Simplicity meets complexity.J Biol Chem. 2019 Feb 8;294(6):2121-2132. doi: 10.1074/jbc.REV118.002809. Epub 2018 Oct 31. J Biol Chem. 2019. PMID: 30385502 Free PMC article. Review.
-
Characterization of a small heat shock protein, Mx Hsp16.6, of Myxococcus xanthus.J Bacteriol. 2005 Aug;187(15):5236-41. doi: 10.1128/JB.187.15.5236-5241.2005. J Bacteriol. 2005. PMID: 16030217 Free PMC article.
-
Thermal-stable proteins of fruit of long-living Sacred Lotus Nelumbo nucifera Gaertn var. China Antique.Trop Plant Biol. 2013 Sep 1;6(2-3):10.1007/s12042-013-9124-2. doi: 10.1007/s12042-013-9124-2. Trop Plant Biol. 2013. PMID: 24363819 Free PMC article.
-
Heat shock proteins in the retina: Focus on HSP70 and alpha crystallins in ganglion cell survival.Prog Retin Eye Res. 2016 May;52:22-46. doi: 10.1016/j.preteyeres.2016.03.001. Epub 2016 Mar 24. Prog Retin Eye Res. 2016. PMID: 27017896 Free PMC article. Review.
-
Proteomic analysis of acidic chaperones, and stress proteins in extreme halophile Halobacterium NRC-1: a comparative proteomic approach to study heat shock response.Proteome Sci. 2006 Apr 19;4:6. doi: 10.1186/1477-5956-4-6. Proteome Sci. 2006. PMID: 16623945 Free PMC article.
References
-
- van den Ijssel, P., Norman, D. G. & Quinlan, R. A. (1999) Curr. Biol. 119 R103-R105. - PubMed
-
- Saibil, H. (2000) Curr. Opin. Struct. Biol. 10 251-258. - PubMed
-
- Clark, J. I. & Muchowski, Z. (2000) Curr. Opin. Struct. Biol. 10 52-59. - PubMed
-
- Kim, K. K., Kim, R. & Kim, S.-H. (1998) Nature 394 595-599. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

