Ubiquitination of alpha-synuclein is not required for formation of pathological inclusions in alpha-synucleinopathies
- PMID: 12819014
- PMCID: PMC1868149
- DOI: 10.1016/s0002-9440(10)63633-4
Ubiquitination of alpha-synuclein is not required for formation of pathological inclusions in alpha-synucleinopathies
Abstract
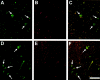
alpha-Synucleinopathies, including Parkinson's disease, dementia with Lewy bodies, and multiple system atrophy, are neurodegenerative disorders in which abnormal inclusions containing alpha-synuclein accumulate in selectively vulnerable neurons and glia. In this report, immunohistochemistry demonstrates ubiquitin in subsets of alpha-synuclein inclusions in dementia with Lewy bodies and multiple system atrophy. Biochemistry demonstrates that alpha-synuclein in the sodium dodecyl sulfate-soluble fractions of diseased brains is ubiquitinated, with mono- and di-ubiquitinated species predominating over polyubiquitinated forms. Similar immunohistochemical and biochemical characteristics were observed in an A53T mutant human alpha-synuclein transgenic mouse model of neurodegenerative alpha-synucleinopathies. Furthermore, in vitro ubiquitination of alpha-synuclein fibrils recapitulated the pattern of alpha-synuclein ubiquitination observed in human disease and the A53T alpha-synuclein mouse model. These results suggest that ubiquitination of alpha-synuclein is not required for inclusion formation and follows the fibrillization of alpha-synuclein.
Figures


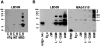

References
-
- Spillantini MG, Goedert M: The α-synucleinopathies: Parkinson’s disease, dementia with Lewy bodies, and multiple system atrophy. Ann NY Acad Sci 2000, 920:16-27 - PubMed
-
- Duda JE, Lee VM-Y, Trojanowski JQ: Neuropathology of synuclein aggregates. J Neurosci Res 2000, 61:121-127 - PubMed
-
- Goedert M: α-Synuclein and neurodegenerative diseases. Nat Rev Neurosci 2001, 2:492-501 - PubMed
-
- Polymeropoulos MH, Lavedan C, Leroy E, Ide SE, Dehejia A, Dutra A, Pike B, Root H, Rubenstein J, Boyer R, Stenroos ES, Chandrasekharappa S, Athanassiadou A, Papapetropoulos T, Johnson WG, Lazzarini AM, Duvoisin RC, Di Iorio G, Golbe LI, Nussbaum RL: Mutation in the α-synuclein gene identified in families with Parkinson’s disease. Science 1997, 276:2045-2047 - PubMed
-
- Kruger R, Kuhn W, Muller T, Woitalla D, Graeber M, Kosel S, Przuntek H, Epplen JT, Schols L, Riess O: Ala30Pro mutation in the gene encoding α-synuclein in Parkinson’s disease. Nat Genet 1998, 18:106-108 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

