Soluble extracts from Helicobacter pylori induce dome formation in polarized intestinal epithelial monolayers in a laminin-dependent manner
- PMID: 12819097
- PMCID: PMC162010
- DOI: 10.1128/IAI.71.7.4067-4078.2003
Soluble extracts from Helicobacter pylori induce dome formation in polarized intestinal epithelial monolayers in a laminin-dependent manner
Abstract
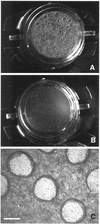
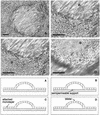
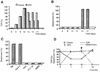
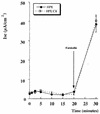
Helicobacter pylori colonizes the stomach at the interface between the mucus layer and the apical pole of gastric epithelial cells. A number of secreted and shed products from the bacteria, such as proteins and lipopolysaccharide, are likely to have a role in the pathogenesis at the epithelial level. To determine the physiological response of transporting polarized epithelia to released soluble factors from the bacterium, we used the T84 cell line. Monolayers of T84 cells were exposed to soluble extracts from H. pylori. The extracts induced rapid "dome" formation as well as an immediate decrease in transepithelial electrical resistance. Domes are fluid-filled blister-like structures unique to polarized epithelia. Their formation has been linked to sodium-transporting events as well as to diminished adherence of the cells to the substrate. H. pylori-induced dome formation in T84 monolayers was exacerbated by amiloride and inhibited by ouabain. Furthermore, it was associated with changes in the expression of the laminin binding alpha 6 beta 4 integrin and the 67-kDa laminin receptor. Domes formed primarily on laminin-coated filters, rather than on fibronectin or collagen matrices, and their formation was inhibited by preincubating the bacterial extract with soluble laminin. This effect was specific to H. pylori and independent of the urease, vacA, cagA, and Lewis phenotype of the strains. These data indicate that released elements from H. pylori can alter the physiological balance and integrity of the epithelium in the absence of an underlying immune response.
Figures





References
-
- Ardini, E., E. Tagliabue, A. Magnifico, S. Buto, V. Castronovo, M. I. Colhaghi, and S. Menard. 1997. Co-regulation and physical association of the 67-kDa monomeric laminin receptor and the α6β4 integrin. J. Biol. Chem. 272:2342-2345. - PubMed
-
- Ashorn, M., F. Cantet, K. Mayo, and F. Megraud. 2000. Cytoskeletal rearrangements induced by Helicobacter pylori strains in epithelial cell culture: possible role of the cytotoxin. Dig. Dis. Sci. 45:1774-1780. - PubMed
-
- Bradford, M. M. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72:248-254. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

