Activation and inhibition of cellular calcium and tyrosine kinase signaling pathways identify targets of the HBx protein involved in hepatitis B virus replication
- PMID: 12829810
- PMCID: PMC161925
- DOI: 10.1128/jvi.77.14.7713-7719.2003
Activation and inhibition of cellular calcium and tyrosine kinase signaling pathways identify targets of the HBx protein involved in hepatitis B virus replication
Abstract
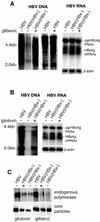
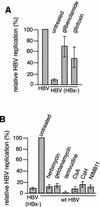
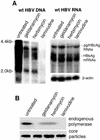
Human hepatitis B virus (HBV) HBx protein is a multifunctional protein that activates cellular signaling pathways and is thought to be essential for viral infection. Woodchuck HBV mutants that lack HBx are unable to replicate in vivo or are severely impaired. HBV replication in HepG2 cells, a human hepatoblastoma cell line, is stimulated 5- to 10-fold by HBx protein. We have utilized the HepG2, HBx-dependent HBV replication system to study the effects of activators and inhibitors of cytosolic calcium and tyrosine kinase signaling pathways on viral replication. By transfecting either a wild-type HBV genome or an HBV genome that does not express HBx and then treating transfected cells with activators or inhibitors of signaling pathways, we identified compounds that either impair wild-type HBV replication or rescue HBx-deficient HBV replication. Geldanamycin or herbimycin A, tyrosine kinase inhibitors, blocked HBV replication. Derivatives of cyclosporine, i.e., cyclosporine A, cyclosporine H, and SDZ NIM811, which block cytosolic calcium signaling and specifically the mitochondrial permeability transition pore (SDZ NIM811), also impaired HBV replication. Treatment of cells with compounds that increase cytosolic calcium levels by a variety of mechanisms rescued replication of an HBx-deficient HBV mutant. Transcription of viral RNA and production of viral capsids were only minimally affected by these treatments. These results define a functional signaling circuit for HBV replication that includes calcium signaling and activation of cytosolic signaling pathways involving Src kinases, and they suggest that these pathways are stimulated by HBx acting on the mitochondrial transition pore.
Figures


Similar articles
-
Calcium signaling by HBx protein in hepatitis B virus DNA replication.Science. 2001 Dec 14;294(5550):2376-8. doi: 10.1126/science.294.5550.2376. Science. 2001. PMID: 11743208
-
Hepatitis B virus replication is associated with an HBx-dependent mitochondrion-regulated increase in cytosolic calcium levels.J Virol. 2007 Nov;81(21):12061-5. doi: 10.1128/JVI.00740-07. Epub 2007 Aug 15. J Virol. 2007. PMID: 17699583 Free PMC article.
-
Activation of focal adhesion kinase by hepatitis B virus HBx protein: multiple functions in viral replication.J Virol. 2006 May;80(9):4406-14. doi: 10.1128/JVI.80.9.4406-4414.2006. J Virol. 2006. PMID: 16611900 Free PMC article.
-
Inhibitory effect of cyclosporine A on hepatitis B virus replication in vitro and its possible mechanisms.Hepatobiliary Pancreat Dis Int. 2005 Feb;4(1):18-22. Hepatobiliary Pancreat Dis Int. 2005. PMID: 15730912 Review.
-
Hepatitis B Virus X and Regulation of Viral Gene Expression.Cold Spring Harb Perspect Med. 2016 Jan 8;6(3):a021402. doi: 10.1101/cshperspect.a021402. Cold Spring Harb Perspect Med. 2016. PMID: 26747833 Free PMC article. Review.
Cited by
-
Operation of mitochondrial machinery in viral infection-induced immune responses.Biochem Pharmacol. 2018 Oct;156:348-356. doi: 10.1016/j.bcp.2018.08.044. Epub 2018 Aug 31. Biochem Pharmacol. 2018. PMID: 30172712 Free PMC article. Review.
-
Dietary calcium is inversely associated with hepatitis B virus infection: an analysis of US National Health and Nutrition Examination Survey (NHANES) 2007-2020.J Health Popul Nutr. 2024 Mar 6;43(1):38. doi: 10.1186/s41043-024-00532-4. J Health Popul Nutr. 2024. PMID: 38449003 Free PMC article.
-
Liver Diseases: From Bench to Bedside.Int J Mol Sci. 2024 May 17;25(10):5454. doi: 10.3390/ijms25105454. Int J Mol Sci. 2024. PMID: 38791491 Free PMC article.
-
Replication of the hepatitis B virus requires a calcium-dependent HBx-induced G1 phase arrest of hepatocytes.Virology. 2010 Nov 10;407(1):14-25. doi: 10.1016/j.virol.2010.07.042. Epub 2010 Aug 16. Virology. 2010. PMID: 20719353 Free PMC article.
-
Hepatitis B and C virus hepatocarcinogenesis: lessons learned and future challenges.Cancer Lett. 2011 Jun 28;305(2):123-43. doi: 10.1016/j.canlet.2010.11.014. Epub 2010 Dec 18. Cancer Lett. 2011. PMID: 21168955 Free PMC article. Review.
References
-
- Andrisani, O., and S. Barnabas. 1999. The transcriptional function of the hepatitis B virus X protein and its role in hepatocarcinogenesis. Int. J. Oncol. 15:1-7. - PubMed
-
- Blagosklonny, M. V. 2002. Hsp-90-associated oncoproteins: multiple targets of geldanamycin and its analogs. Leukemia 16:455-462. - PubMed
-
- Bouchard, M. J., L. H. Wang, and R. J. Schneider. 2001. Calcium signaling by HBx protein in hepatitis B virus DNA replication. Science 294:2376-2378. - PubMed
-
- Clapham, D. E. 1997. Calcium signaling. Cell 80:259-268. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

