The human multidrug resistance protein MRP4 functions as a prostaglandin efflux transporter and is inhibited by nonsteroidal antiinflammatory drugs
- PMID: 12835412
- PMCID: PMC170903
- DOI: 10.1073/pnas.1033060100
The human multidrug resistance protein MRP4 functions as a prostaglandin efflux transporter and is inhibited by nonsteroidal antiinflammatory drugs
Abstract
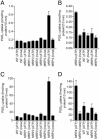
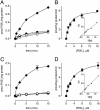
Prostaglandins are involved in a wide variety of physiological and pathophysiological processes, but the mechanism of prostaglandin release from cells is not completely understood. Although poorly membrane permeable, prostaglandins are believed to exit cells by passive diffusion. We have investigated the interaction between prostaglandins and members of the ATP-binding cassette (ABC) transporter ABCC [multidrug resistance protein (MRP)] family of membrane export pumps. In inside-out membrane vesicles derived from insect cells or HEK293 cells, MRP4 catalyzed the time- and ATP-dependent uptake of prostaglandin E1 (PGE1) and PGE2. In contrast, MRP1, MRP2, MRP3, and MRP5 did not transport PGE1 or PGE2. The MRP4-mediated transport of PGE1 and PGE2 displayed saturation kinetics, with Km values of 2.1 and 3.4 microM, respectively. Further studies showed that PGF1alpha, PGF2alpha, PGA1, and thromboxane B2 were high-affinity inhibitors (and therefore presumably substrates) of MRP4. Furthermore, several nonsteroidal antiinflammatory drugs were potent inhibitors of MRP4 at concentrations that did not inhibit MRP1. In cells expressing the prostaglandin transporter PGT, the steady-state accumulation of PGE1 and PGE2 was reduced proportional to MRP4 expression. Inhibition of MRP4 by an MRP4-specific RNA interference construct or by indomethacin reversed this accumulation deficit. Together, these data suggest that MRP4 can release prostaglandins from cells, and that, in addition to inhibiting prostaglandin synthesis, some nonsteroidal antiinflammatory drugs might also act by inhibiting this release.
Figures


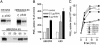
Comment in
-
Nonsteroidal antiinflammatory drugs inhibiting prostanoid efflux: as easy as ABC?Proc Natl Acad Sci U S A. 2003 Aug 5;100(16):9108-10. doi: 10.1073/pnas.1733826100. Epub 2003 Jul 28. Proc Natl Acad Sci U S A. 2003. PMID: 12886006 Free PMC article. No abstract available.
Similar articles
-
Multidrug resistance protein 4 (ABCC4)-mediated ATP hydrolysis: effect of transport substrates and characterization of the post-hydrolysis transition state.J Biol Chem. 2004 Nov 19;279(47):48855-64. doi: 10.1074/jbc.M408849200. Epub 2004 Sep 9. J Biol Chem. 2004. PMID: 15364914
-
Prostanoid transport by multidrug resistance protein 4 (MRP4/ABCC4) localized in tissues of the human urogenital tract.J Urol. 2005 Dec;174(6):2409-14. doi: 10.1097/01.ju.0000180411.03808.cb. J Urol. 2005. PMID: 16280858
-
The multidrug resistance-associated protein 4 (MRP4) appears as a functional carrier of prostaglandins regulated by oxytocin in the bovine endometrium.Endocrinology. 2011 Dec;152(12):4993-5004. doi: 10.1210/en.2011-1406. Epub 2011 Oct 11. Endocrinology. 2011. PMID: 21990316
-
Substrates and inhibitors of human multidrug resistance associated proteins and the implications in drug development.Curr Med Chem. 2008;15(20):1981-2039. doi: 10.2174/092986708785132870. Curr Med Chem. 2008. PMID: 18691054 Review.
-
Multidrug resistance-associated proteins: Export pumps for conjugates with glutathione, glucuronate or sulfate.Biofactors. 2003;17(1-4):103-14. doi: 10.1002/biof.5520170111. Biofactors. 2003. PMID: 12897433 Review.
Cited by
-
Intersecting Mechanisms of Hypoxia and Prostaglandin E2-Mediated Inflammation in the Comparative Biology of Oral Squamous Cell Carcinoma.Front Oncol. 2021 May 21;11:539361. doi: 10.3389/fonc.2021.539361. eCollection 2021. Front Oncol. 2021. PMID: 34094895 Free PMC article. Review.
-
Pharmacogenetics of chronic pain and its treatment.Mediators Inflamm. 2013;2013:864319. doi: 10.1155/2013/864319. Epub 2013 May 20. Mediators Inflamm. 2013. PMID: 23766564 Free PMC article. Review.
-
Dual role of cAMP in the transcriptional regulation of multidrug resistance-associated protein 4 (MRP4) in pancreatic adenocarcinoma cell lines.PLoS One. 2015 Mar 19;10(3):e0120651. doi: 10.1371/journal.pone.0120651. eCollection 2015. PLoS One. 2015. PMID: 25790437 Free PMC article.
-
Synthesis and Significance of Arachidonic Acid, a Substrate for Cyclooxygenases, Lipoxygenases, and Cytochrome P450 Pathways in the Tumorigenesis of Glioblastoma Multiforme, Including a Pan-Cancer Comparative Analysis.Cancers (Basel). 2023 Feb 2;15(3):946. doi: 10.3390/cancers15030946. Cancers (Basel). 2023. PMID: 36765904 Free PMC article. Review.
-
Role of the efflux transporters BCRP and MRP1 in human placental bio-disposition of pravastatin.Biochem Pharmacol. 2018 Oct;156:467-478. doi: 10.1016/j.bcp.2018.09.012. Epub 2018 Sep 12. Biochem Pharmacol. 2018. PMID: 30217571 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

