The acute neurotoxicity of mefloquine may be mediated through a disruption of calcium homeostasis and ER function in vitro
- PMID: 12848898
- PMCID: PMC194860
- DOI: 10.1186/1475-2875-2-14
The acute neurotoxicity of mefloquine may be mediated through a disruption of calcium homeostasis and ER function in vitro
Abstract
Background: There is no established biochemical basis for the neurotoxicity of mefloquine. We investigated the possibility that the acute in vitro neurotoxicity of mefloquine might be mediated through a disruptive effect of the drug on endoplasmic reticulum (ER) calcium homeostasis.
Methods: Laser scanning confocal microscopy was employed to monitor real-time changes in basal intracellular calcium concentrations in embryonic rat neurons in response to mefloquine and thapsigargin (a known inhibitor of the ER calcium pump) in the presence and absence of external calcium. Changes in the transcriptional regulation of known ER stress response genes in neurons by mefloquine were investigated using Affymetrix arrays. The MTT assay was employed to measure the acute neurotoxicity of mefloquine and its antagonisation by thapsigargin.
Results: At physiologically relevant concentrations mefloquine was found to mobilize neuronal ER calcium stores and antagonize the pharmacological action of thapsigargin, a specific inhibitor of the ER calcium pump. Mefloquine also induced a sustained influx of extra-neuronal calcium via an unknown mechanism. The transcription of key ER proteins including GADD153, PERK, GRP78, PDI, GRP94 and calreticulin were up-regulated by mefloquine, suggesting that the drug induced an ER stress response. These effects appear to be related, in terms of dose effect and kinetics of action, to the acute neurotoxicity of the drug in vitro.
Conclusions: Mefloquine was found to disrupt neuronal calcium homeostasis and induce an ER stress response at physiologically relevant concentrations, effects that may contribute, at least in part, to the neurotoxicity of the drug in vitro.
Figures

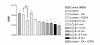
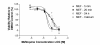

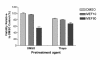
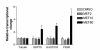
Similar articles
-
Transcriptional profiling of mefloquine-induced disruption of calcium homeostasis in neurons in vitro.Genomics. 2005 Nov;86(5):539-50. doi: 10.1016/j.ygeno.2005.07.004. Epub 2005 Aug 18. Genomics. 2005. PMID: 16109470
-
The endoplasmic reticulum-related events in S-nitrosoglutathione-induced neurotoxicity in cerebellar granule cells.Brain Res. 2004 Jul 23;1015(1-2):25-33. doi: 10.1016/j.brainres.2004.04.022. Brain Res. 2004. PMID: 15223363
-
Effect of nitric oxide on endoplasmic reticulum calcium homeostasis, protein synthesis and energy metabolism.Cell Calcium. 2000 Feb;27(2):107-15. doi: 10.1054/ceca.1999.0099. Cell Calcium. 2000. PMID: 10756977
-
Disturbances of the functioning of endoplasmic reticulum: a key mechanism underlying neuronal cell injury?J Cereb Blood Flow Metab. 1999 Jan;19(1):1-18. doi: 10.1097/00004647-199901000-00001. J Cereb Blood Flow Metab. 1999. PMID: 9886350 Review.
-
Dual effects of neuroprotection and neurotoxicity by general anesthetics: role of intracellular calcium homeostasis.Prog Neuropsychopharmacol Biol Psychiatry. 2013 Dec 2;47:156-61. doi: 10.1016/j.pnpbp.2013.05.009. Epub 2013 May 28. Prog Neuropsychopharmacol Biol Psychiatry. 2013. PMID: 23721657 Free PMC article. Review.
Cited by
-
Neurological and psychiatric safety of tafenoquine in Plasmodium vivax relapse prevention: a review.Malar J. 2020 Mar 14;19(1):111. doi: 10.1186/s12936-020-03184-x. Malar J. 2020. PMID: 32169086 Free PMC article. Review.
-
Mefloquine neurotoxicity is mediated by non-receptor tyrosine kinase.Neurotoxicology. 2011 Oct;32(5):578-85. doi: 10.1016/j.neuro.2011.01.001. Epub 2011 Jan 15. Neurotoxicology. 2011. PMID: 21241737 Free PMC article.
-
Anti-malaria drug mefloquine induces motor learning deficits in humans.Front Neurosci. 2010 Nov 19;4:191. doi: 10.3389/fnins.2010.00191. eCollection 2010. Front Neurosci. 2010. PMID: 21151372 Free PMC article.
-
Prediction of improved antimalarial chemotherapy of artesunate-mefloquine in combination with mefloquine sensitive and resistant Plasmodium falciparum malaria.PLoS One. 2023 Feb 23;18(2):e0282099. doi: 10.1371/journal.pone.0282099. eCollection 2023. PLoS One. 2023. PMID: 36821622 Free PMC article.
-
Mefloquine effects on ventral tegmental area dopamine and GABA neuron inhibition: a physiologic role for connexin-36 GAP junctions.Synapse. 2011 Aug;65(8):804-13. doi: 10.1002/syn.20907. Epub 2011 Apr 7. Synapse. 2011. PMID: 21218452 Free PMC article.
References
-
- Phillips-Howard PA, ter Kuile FO. CNS adverse events associated with antimalarial agents: Fact or fiction? Drug Safety. 1995;12:370–383. - PubMed
-
- Schlagenhauf P. Mefloquine for malaria chemoprophylaxis 1992–1998. J Travel Med. 1999;6:122–123. - PubMed
-
- Kollaritsch H, Karbwang J, Wiedermann G, Mikolasek A, Na-Bangchang K, Wernsdorfer WH. Mefloquine concentration profiles during prophylactic dose regimens. Wien Klin Wochenschr. 2000;112:441–7. - PubMed
-
- Simpson JA, Price R, ter Kuile F, Teja-Isavatharm P, Nosten F, Chongsuphajaisiddhi T, Looareesuwan S, Aarons L, White NJ. Population pharmacokinetics of mefloquine in patients with acute falciparum malaria. Clin Pharmacol Ther. 1999;66:472–84. - PubMed
-
- Lee HS, Go ML. Effects of mefloquine on Ca2+ uptake and release by dog brain microsomes. Arch Int Pharmacodyn Ther. 1995;331:221–231. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

