Protein kinase A phosphorylation modulates transport of the polypyrimidine tract-binding protein
- PMID: 12851456
- PMCID: PMC166389
- DOI: 10.1073/pnas.1432696100
Protein kinase A phosphorylation modulates transport of the polypyrimidine tract-binding protein
Abstract
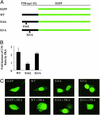
The heterogeneous nuclear ribonucleoprotein particle (hnRNP) proteins play important roles in mRNA processing in eukaryotes, but little is known about how they are regulated by cellular signaling pathways. The polypyrimidine-tract binding protein (PTB, or hnRNP I) is an important regulator of alternative pre-mRNA splicing, of viral RNA translation, and of mRNA localization. Here we show that the nucleo-cytoplasmic transport of PTB is regulated by the 3',5'-cAMP-dependent protein kinase (PKA). PKA directly phosphorylates PTB on conserved Ser-16, and PKA activation in PC12 cells induces Ser-16 phosphorylation. PTB carrying a Ser-16 to alanine mutation accumulates normally in the nucleus. However, export of this mutant protein from the nucleus is greatly reduced in heterokaryon shuttling assays. Conversely, hyperphosphorylation of PTB by coexpression with the catalytic subunit of PKA results in the accumulation of PTB in the cytoplasm. This accumulation is again specifically blocked by the S16A mutation. Similarly, in Xenopus oocytes, the phospho-Ser-16-PTB is restricted to the cytoplasm, whereas the non-Ser-16-phosphorylated PTB is nuclear. Thus, direct PKA phosphorylation of PTB at Ser-16 modulates the nucleo-cytoplasmic distribution of PTB. This phosphorylation likely plays a role in the cytoplasmic function of PTB.
Figures


Similar articles
-
Relocalization of the polypyrimidine tract-binding protein during PKA-induced neurite growth.Biochim Biophys Acta. 2007 Jun;1773(6):912-23. doi: 10.1016/j.bbamcr.2007.02.006. Epub 2007 Feb 22. Biochim Biophys Acta. 2007. PMID: 17400307
-
Human polypyrimidine tract-binding protein interacts with mitochondrial tRNA(Thr) in the cytosol.Nucleic Acids Res. 2016 Feb 18;44(3):1342-53. doi: 10.1093/nar/gkv1355. Epub 2015 Dec 10. Nucleic Acids Res. 2016. PMID: 26657638 Free PMC article.
-
Cytoplasmic translocation of polypyrimidine tract-binding protein and its binding to viral RNA during Japanese encephalitis virus infection inhibits virus replication.PLoS One. 2014 Dec 29;9(12):e114931. doi: 10.1371/journal.pone.0114931. eCollection 2014. PLoS One. 2014. PMID: 25545659 Free PMC article.
-
Polypyrimidine-tract-binding protein: a multifunctional RNA-binding protein.Biochem Soc Trans. 2008 Aug;36(Pt 4):641-7. doi: 10.1042/BST0360641. Biochem Soc Trans. 2008. PMID: 18631133 Review.
-
PTB: Not just a polypyrimidine tract-binding protein.J Cell Physiol. 2022 May;237(5):2357-2373. doi: 10.1002/jcp.30716. Epub 2022 Mar 14. J Cell Physiol. 2022. PMID: 35288937 Review.
Cited by
-
Splicing factor arginine/serine-rich 17A (SFRS17A) is an A-kinase anchoring protein that targets protein kinase A to splicing factor compartments.J Biol Chem. 2009 Dec 11;284(50):35154-64. doi: 10.1074/jbc.M109.056465. Epub 2009 Oct 19. J Biol Chem. 2009. PMID: 19840947 Free PMC article.
-
Knockdown of splicing factor SRp20 causes apoptosis in ovarian cancer cells and its expression is associated with malignancy of epithelial ovarian cancer.Oncogene. 2011 Jan 20;30(3):356-65. doi: 10.1038/onc.2010.426. Epub 2010 Sep 20. Oncogene. 2011. PMID: 20856201 Free PMC article.
-
A conserved serine of heterogeneous nuclear ribonucleoprotein L (hnRNP L) mediates depolarization-regulated alternative splicing of potassium channels.J Biol Chem. 2012 Jun 29;287(27):22709-16. doi: 10.1074/jbc.M112.357343. Epub 2012 May 8. J Biol Chem. 2012. PMID: 22570490 Free PMC article.
-
Remodelling of a polypyrimidine tract-binding protein complex during apoptosis activates cellular IRESs.Cell Death Differ. 2014 Jan;21(1):161-71. doi: 10.1038/cdd.2013.135. Epub 2013 Oct 18. Cell Death Differ. 2014. PMID: 24141718 Free PMC article.
-
Activation of alpha-tropomyosin exon 2 is regulated by the SR protein 9G8 and heterogeneous nuclear ribonucleoproteins H and F.Mol Cell Biol. 2006 Dec;26(23):8791-802. doi: 10.1128/MCB.01677-06. Epub 2006 Sep 25. Mol Cell Biol. 2006. PMID: 17000773 Free PMC article.
References
-
- Krecic, A. M. & Swanson, M. S. (1999) Curr. Opin. Cell Biol. 11, 363–371. - PubMed
-
- Mattaj, I. W. & Englmeier, L. (1998) Annu. Rev. Biochem. 67, 265–306. - PubMed
-
- Nakielny, S. & Dreyfuss, G. (1999) Cell 99, 677–690. - PubMed
-
- Habelhah, H., Shah, K., Huang, L., Ostareck-Lederer, A., Burlingame, A. L., Shokat, K. M., Hentze, M. W. & Ronai, Z. E. (2001) Nat. Cell Biol. 3, 325–330. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

