Cav1.4alpha1 subunits can form slowly inactivating dihydropyridine-sensitive L-type Ca2+ channels lacking Ca2+-dependent inactivation
- PMID: 12853422
- PMCID: PMC6740341
- DOI: 10.1523/JNEUROSCI.23-14-06041.2003
Cav1.4alpha1 subunits can form slowly inactivating dihydropyridine-sensitive L-type Ca2+ channels lacking Ca2+-dependent inactivation
Abstract
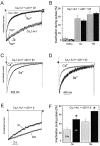
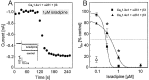
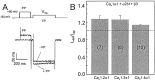
The neuronal L-type calcium channels (LTCCs) Cav1.2alpha1 and Cav1.3alpha1 are functionally distinct. Cav1.3alpha1 activates at lower voltages and inactivates more slowly than Cav1.2alpha1, making it suitable to support sustained L-type Ca2+ inward currents (ICa,L) and serve in pacemaker functions. We compared the biophysical and pharmacological properties of human retinal Cav1.4alpha1 using the whole-cell patch-clamp technique after heterologous expression in tsA-201 cells with other L-type alpha1 subunits. Cav1.4alpha1-mediated inward Ba2+ currents (IBa) required the coexpression of alpha2delta1 and beta3 or beta2a subunits and were detected in a lower proportion of transfected cells than Cav1.3alpha1. IBa activated at more negative voltages (5% activation threshold; -39mV; 15 mm Ba2+) than Cav1.2alpha1 and slightly more positive than Cav1.3alpha1. Voltage-dependent inactivation of IBa was slower than for Cav1.2alpha1 and Cav1.3alpha1( approximately 50% inactivation after 5 sec; alpha2delta1 + beta3 coexpression). Inactivation was not increased with Ca2+ as the charge carrier, indicating the absence of Ca2+-dependent inactivation. Cav1.4alpha1 exhibited voltage-dependent, G-protein-independent facilitation by strong depolarizing pulses. The dihydropyridine (DHP)-antagonist isradipine blocked Cav1.4alpha1 with approximately 15-fold lower sensitivity than Cav1.2alpha1 and in a voltage-dependent manner. Strong stimulation by the DHP BayK 8644 was found despite the substitution of an otherwise L-type channel-specific tyrosine residue in position 1414 (repeat IVS6) by a phenylalanine. Cav1.4alpha1 + alpha2delta1 + beta channel complexes can form LTCCs with intermediate DHP antagonist sensitivity lacking Ca2+-dependent inactivation. Their biophysical properties should enable them to contribute to sustained ICa,L at negative potentials, such as required for tonic neurotransmitter release in sensory cells and plateau potentials in spiking neurons.
Figures





Similar articles
-
Functional characterization of the L-type Ca2+ channel Cav1.4alpha1 from mouse retina.Invest Ophthalmol Vis Sci. 2004 Feb;45(2):708-13. doi: 10.1167/iovs.03-0937. Invest Ophthalmol Vis Sci. 2004. PMID: 14744918
-
alpha 1D (Cav1.3) subunits can form l-type Ca2+ channels activating at negative voltages.J Biol Chem. 2001 Jun 22;276(25):22100-6. doi: 10.1074/jbc.M101469200. Epub 2001 Apr 2. J Biol Chem. 2001. PMID: 11285265
-
Functional characterization of ion permeation pathway in the N-type Ca2+ channel.J Neurophysiol. 1998 Feb;79(2):622-34. doi: 10.1152/jn.1998.79.2.622. J Neurophysiol. 1998. PMID: 9463426
-
Voltage-dependent calcium channels.Gen Physiol Biophys. 2005 Jun;24 Suppl 1:1-78. Gen Physiol Biophys. 2005. PMID: 16096350 Review.
-
L-type CaV1.2 calcium channels: from in vitro findings to in vivo function.Physiol Rev. 2014 Jan;94(1):303-26. doi: 10.1152/physrev.00016.2013. Physiol Rev. 2014. PMID: 24382889 Review.
Cited by
-
Location of release sites and calcium-activated chloride channels relative to calcium channels at the photoreceptor ribbon synapse.J Neurophysiol. 2011 Jan;105(1):321-35. doi: 10.1152/jn.00332.2010. Epub 2010 Nov 17. J Neurophysiol. 2011. PMID: 21084687 Free PMC article.
-
Mosaic synaptopathy and functional defects in Cav1.4 heterozygous mice and human carriers of CSNB2.Hum Mol Genet. 2014 Mar 15;23(6):1538-50. doi: 10.1093/hmg/ddt541. Epub 2013 Oct 26. Hum Mol Genet. 2014. PMID: 24163243 Free PMC article.
-
Mutation associated with an autosomal dominant cone-rod dystrophy CORD7 modifies RIM1-mediated modulation of voltage-dependent Ca2+ channels.Channels (Austin). 2007 May-Jun;1(3):144-7. doi: 10.4161/chan.4660. Epub 2007 Jun 29. Channels (Austin). 2007. PMID: 18690027 Free PMC article.
-
Cav1.4 congenital stationary night blindness is associated with an increased rate of proteasomal degradation.Front Cell Dev Biol. 2023 May 3;11:1161548. doi: 10.3389/fcell.2023.1161548. eCollection 2023. Front Cell Dev Biol. 2023. PMID: 37206923 Free PMC article.
-
The Contribution of L-Type Cav1.3 Channels to Retinal Light Responses.Front Mol Neurosci. 2017 Dec 5;10:394. doi: 10.3389/fnmol.2017.00394. eCollection 2017. Front Mol Neurosci. 2017. PMID: 29259539 Free PMC article.
References
-
- Alaburda A, Perrier JF, Hounsgaard J ( 2002) Mechanisms causing plateau potentials in spinal motoneurones. Adv Exp Med Biol 508: 219–226. - PubMed
-
- Ball SL, Powers PA, Shin HS, Morgans CW, Peachey NS, Gregg RG ( 2002) Role of the beta(2) subunit of voltage-dependent calcium channels in the retinal outer plexiform layer. Invest Ophthalmol Vis Sci 43: 1595–1603. - PubMed
-
- Bech-Hansen NT, Naylor MJ, Maybaum TA, Pearce WG, Koop B, Fishman GA, Mets M, Musarella MA, Boycott KM ( 1998) Loss-of-function mutations in a calcium-channel alpha1-subunit gene in Xp11.23 cause incomplete X-linked congenital stationary night blindness. Nat Genet 19: 264–267. - PubMed
-
- Berntson A, Taylor WR, Morgans CW ( 2003) Molecular identity, synaptic localization, and physiology of calcium channels in retinal bipolar cells. J Neurosci Res 71: 146–151. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous
