Presynaptic depression of glutamatergic synaptic transmission by D1-like dopamine receptor activation in the avian basal ganglia
- PMID: 12853427
- PMCID: PMC6740340
- DOI: 10.1523/JNEUROSCI.23-14-06086.2003
Presynaptic depression of glutamatergic synaptic transmission by D1-like dopamine receptor activation in the avian basal ganglia
Abstract
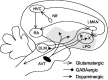
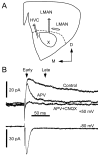
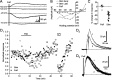
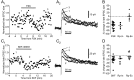
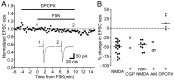
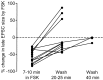
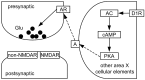
Vocal behavior in songbirds exemplifies a rich integration of motor, cognitive, and social functions that are shared among vertebrates. As a part of the underlying neural substrate, the song system, the anterior forebrain pathway (AFP) is required for song learning and maintenance. The AFP resembles the mammalian basal ganglia-thalamocortical loop in its macroscopic organization, neuronal intrinsic properties, and microcircuitry. Area X, the first station in the AFP, is a part of the basal ganglia essential for vocal learning. It receives glutamatergic inputs from pallial structures and sends GABAergic outputs to thalamic structures. It also receives dense dopaminergic innervation from the midbrain. The role of this innervation is essentially unknown. Here we provide evidence that dopamine (DA) can modulate the glutamatergic inputs to spiny neurons in area X. In whole-cell voltage-clamp recordings from neurons in brain slices of adult zebra finches, we found that activation of D1-like DA receptors depresses ionotropic glutamatergic synaptic current in area X spiny neurons. This effect is mediated by a presynaptic site of action, mimicked by activation of adenylyl cyclase, and blocked by protein kinase A inhibitor and an adenosine A1 receptor antagonist. These results suggest that, in addition to altering the input-output function of spiny neurons by modulating their excitability, as we have shown previously, DA can directly influence the excitatory inputs to these neurons as well. Thus, DA can exert fine control over information processing through spiny neurons in area X, the dynamics of the AFP output, and ultimately song learning and maintenance.
Figures




Similar articles
-
Anatomy of a songbird basal ganglia circuit essential for vocal learning and plasticity.J Chem Neuroanat. 2010 Mar;39(2):124-31. doi: 10.1016/j.jchemneu.2009.07.003. Epub 2009 Jul 22. J Chem Neuroanat. 2010. PMID: 19596062 Free PMC article. Review.
-
Dopamine modulates excitability of spiny neurons in the avian basal ganglia.J Neurosci. 2002 Jun 15;22(12):5210-8. doi: 10.1523/JNEUROSCI.22-12-05210.2002. J Neurosci. 2002. PMID: 12077216 Free PMC article.
-
Endocannabinoids mediate synaptic plasticity at glutamatergic synapses on spiny neurons within a basal ganglia nucleus necessary for song learning.J Neurophysiol. 2011 Mar;105(3):1159-69. doi: 10.1152/jn.00676.2010. Epub 2010 Dec 22. J Neurophysiol. 2011. PMID: 21177997 Free PMC article.
-
Long-term potentiation in an avian basal ganglia nucleus essential for vocal learning.J Neurosci. 2004 Jan 14;24(2):488-94. doi: 10.1523/JNEUROSCI.4358-03.2004. J Neurosci. 2004. PMID: 14724247 Free PMC article.
-
Social modulation of learned behavior by dopamine in the basal ganglia: insights from songbirds.J Physiol Paris. 2013 Jun;107(3):219-29. doi: 10.1016/j.jphysparis.2012.09.002. Epub 2012 Sep 29. J Physiol Paris. 2013. PMID: 23032272 Review.
Cited by
-
Dopamine Modulates Excitatory Synaptic Transmission by Activating Presynaptic D1-like Dopamine Receptors in the RA Projection Neurons of Zebra Finches.Front Cell Neurosci. 2020 May 12;14:126. doi: 10.3389/fncel.2020.00126. eCollection 2020. Front Cell Neurosci. 2020. PMID: 32477072 Free PMC article.
-
Neurochemical Characterization of Dopaminoceptive Cells in Song Control Nuclei of Canaries and Their Activation During Song Production: A Multiplex Fluorescent In Situ Hybridization Study.J Comp Neurol. 2024 Oct;532(10):e25675. doi: 10.1002/cne.25675. J Comp Neurol. 2024. PMID: 39387367
-
Anatomy of a songbird basal ganglia circuit essential for vocal learning and plasticity.J Chem Neuroanat. 2010 Mar;39(2):124-31. doi: 10.1016/j.jchemneu.2009.07.003. Epub 2009 Jul 22. J Chem Neuroanat. 2010. PMID: 19596062 Free PMC article. Review.
-
Regulation of prefrontal excitatory neurotransmission by dopamine in the nucleus accumbens core.J Physiol. 2012 Aug 15;590(16):3743-69. doi: 10.1113/jphysiol.2012.235200. Epub 2012 May 14. J Physiol. 2012. PMID: 22586226 Free PMC article.
-
Neurogenesis in the adult avian song-control system.Cold Spring Harb Perspect Biol. 2015 Jun 1;7(6):a019000. doi: 10.1101/cshperspect.a019000. Cold Spring Harb Perspect Biol. 2015. PMID: 26032719 Free PMC article. Review.
References
-
- Blanton MG, Lo Turco JJ, Kriegstein AR ( 1989) Whole cell recording from neurons in slices of reptilian and mammalian cerebral cortex. J Neurosci Methods 30: 203–210. - PubMed
-
- Bonci A, Williams JT ( 1996) A common mechanism mediates long-term changes in synaptic transmission after chronic cocaine and morphine. Neuron 16: 631–639. - PubMed
-
- Bottjer SW ( 1993) The distribution of tyrosine hydroxylase immunoreactivity in the brains of male and female zebra finches. J Neurobiol 24: 51–69. - PubMed
-
- Bottjer SW, Johnson F ( 1997) Circuits, hormones, and learning: vocal behavior in songbirds. J Neurobiol 33: 602–618. - PubMed
-
- Bottjer SW, Miesner EA, Arnold AP ( 1984) Forebrain lesions disrupt development but not maintenance of song in passerine birds. Science 224: 901–903. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
