Selective interactions of human kin17 and RPA proteins with chromatin and the nuclear matrix in a DNA damage- and cell cycle-regulated manner
- PMID: 12853634
- PMCID: PMC165974
- DOI: 10.1093/nar/gkg459
Selective interactions of human kin17 and RPA proteins with chromatin and the nuclear matrix in a DNA damage- and cell cycle-regulated manner
Abstract
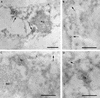
Several proteins involved in DNA synthesis are part of the so-called 'replication factories' that are anchored on non-chromatin nuclear structures. We report here that human kin17, a nuclear stress-activated protein, associates with both chromatin and non-chromatin nuclear structures in a cell cycle- and DNA damage-dependent manner. After L-mimosine block and withdrawal we observed that kin17 protein was recruited in the nucleus during re-entry and progression through S phase. These results are consistent with a role of kin17 protein in DNA replication. About 50% of the total amount of kin17 protein was detected on nuclear structures and could not be released by detergents. Furthermore, the amount of kin17 protein greatly increased in both G(1)/S and S phase-arrested cells in fractions containing proteins anchored to nuclear structures. The detection of kin17 protein showed for the first time its preferential assembly within non-chromatin nuclear structures in G(1)/S and S phase-arrested cells, while the association with these structures was found to be less stable in the G(2)/M phase, as judged by fractionation of human cells and immunostaining. In asynchronous growing cells, kin17 protein interacted with both chromatin DNA and non-chromatin nuclear structures, while in S phase-arrested cells it interacted mostly with non-chromatin nuclear structures, as judged by DNase I treatment and in vivo UV cross-linking. In the presence of DNA damage in S phase cells, the distribution of kin17 protein became mainly associated with chromosomal DNA, as judged by limited formaldehyde cross-linking of living cells. The physical interaction of kin17 protein with components of the nuclear matrix was confirmed and visualized by indirect immunofluorescence and immunoelectron microscopy. Our results indicate that, during S phase, a fraction of the human kin17 protein preferentially associates with the nuclear matrix, a fundamentally non-chromatin higher order nuclear structure, and to chromatin DNA in the presence of DNA damage.
Figures






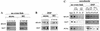
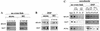
References
-
- Boulikas T. (1996) Common structural features of replication origins in all life forms. J. Cell. Biochem., 60, 297–388. - PubMed
-
- Boulikas T. (1995) Chromatin domains and the prediction of MAR sequences. Int. Rev. Cytol., 162A, 279–388. - PubMed
-
- Cook P.R. (1988) The nucleoskeleton: artefact, passive framework or active site? J. Cell Sci., 90, 1–6. - PubMed
-
- Berezney R., Mortillaro,M.J., Ma,H., Wei,X. and Samarabandu,J. (1995) The nuclear matrix: a structural milieu for genomic function. Int. Rev. Cytol., 162A, 1–65. - PubMed
-
- Nickerson J.A. (2001) Experimental observations of a nuclear matrix. J. Cell Sci., 114, 463–474. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

