Improved protection of rhesus macaques against intrarectal simian immunodeficiency virus SIV(mac251) challenge by a replication-competent Ad5hr-SIVenv/rev and Ad5hr-SIVgag recombinant priming/gp120 boosting regimen
- PMID: 12857905
- PMCID: PMC165263
- DOI: 10.1128/jvi.77.15.8354-8365.2003
Improved protection of rhesus macaques against intrarectal simian immunodeficiency virus SIV(mac251) challenge by a replication-competent Ad5hr-SIVenv/rev and Ad5hr-SIVgag recombinant priming/gp120 boosting regimen
Abstract
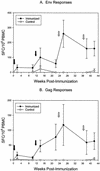
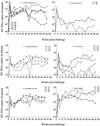
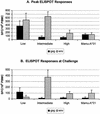
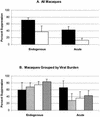
In this study we investigated the ability of a replication-competent Ad5hr-SIVenv/rev and Ad5hr-SIVgag recombinant priming/gp120 boosting regimen to induce protective immunity in rhesus macaques against pathogenic simian immunodeficiency virus(mac251). Immunization of macaques by two sequential administrations of the same recombinants by the same route resulted in boosting and persistence of SIV-specific cellular immune responses for 42 weeks past the initial immunization. Anti-SIV gp120 immunoglobulin G (IgG) and IgA antibodies were induced in secretory fluids, and all macaques exhibited serum neutralizing antibody activity. After intrarectal SIV(mac251) challenge, all of the macaques became infected. However, relative protection, as assessed by statistically significant lower SIV viral loads in plasma at both acute infection and set point, was observed in 8 out of 12 immunized non-Mamu-A(*)01 animals. Elevated mean cellular immune responses to Gag and Env, neutralizing antibody activity, and IgG and IgA binding antibody levels were observed in the eight protected macaques. Statistically significant correlations with protective outcome were observed for cellular immune responses to SIV Env and Gag and for SIV gp120-specific IgG antibodies in nasal and vaginal fluids. Two macaques that exhibited the greatest and most persistent viremia control also exhibited strong CD8(+) T-cell antiviral activity. The results suggest that a spectrum of immune responses may be necessary for adequate control of viral replication and disease progression and highlight a potential role for nonneutralizing antibodies at mucosal sites.
Figures



Similar articles
-
A replication competent adenovirus 5 host range mutant-simian immunodeficiency virus (SIV) recombinant priming/subunit protein boosting vaccine regimen induces broad, persistent SIV-specific cellular immunity to dominant and subdominant epitopes in Mamu-A*01 rhesus macaques.J Immunol. 2003 Apr 15;170(8):4281-9. doi: 10.4049/jimmunol.170.8.4281. J Immunol. 2003. PMID: 12682263
-
Evaluation of combination DNA/replication-competent Ad-SIV recombinant immunization regimens in rhesus macaques.AIDS Res Hum Retroviruses. 2004 Feb;20(2):235-44. doi: 10.1089/088922204773004969. AIDS Res Hum Retroviruses. 2004. PMID: 15018712
-
Boosting of SIV-specific immune responses in rhesus macaques by repeated administration of Ad5hr-SIVenv/rev and Ad5hr-SIVgag recombinants.Vaccine. 2003 Sep 8;21(25-26):4022-35. doi: 10.1016/s0264-410x(03)00266-4. Vaccine. 2003. PMID: 12922139
-
Antiviral CD8+ T cells in the genital tract control viral replication and delay progression to AIDS after vaginal SIV challenge in rhesus macaques immunized with virulence attenuated SHIV 89.6.J Intern Med. 2009 Jan;265(1):67-77. doi: 10.1111/j.1365-2796.2008.02051.x. J Intern Med. 2009. PMID: 19093961 Free PMC article. Review.
-
Cannabinoid administration attenuates the progression of simian immunodeficiency virus.AIDS Res Hum Retroviruses. 2011 Jun;27(6):585-92. doi: 10.1089/aid.2010.0218. Epub 2010 Nov 23. AIDS Res Hum Retroviruses. 2011. PMID: 20874519 Free PMC article. Review.
Cited by
-
Obstacles to the successful development of an efficacious T cell-inducing HIV-1 vaccine.J Leukoc Biol. 2009 Oct;86(4):779-93. doi: 10.1189/jlb.0209094. Epub 2009 Jul 13. J Leukoc Biol. 2009. PMID: 19597003 Free PMC article. Review.
-
Protection against mucosal simian immunodeficiency virus SIV(mac251) challenge by using replicating adenovirus-SIV multigene vaccine priming and subunit boosting.J Virol. 2004 Mar;78(5):2212-21. doi: 10.1128/jvi.78.5.2212-2221.2004. J Virol. 2004. PMID: 14963117 Free PMC article.
-
Potent, persistent induction and modulation of cellular immune responses in rhesus macaques primed with Ad5hr-simian immunodeficiency virus (SIV) env/rev, gag, and/or nef vaccines and boosted with SIV gp120.J Virol. 2003 Aug;77(16):8607-20. doi: 10.1128/jvi.77.16.8607-8620.2003. J Virol. 2003. PMID: 12885879 Free PMC article.
-
Systemic neutralizing antibodies induced by long interval mucosally primed systemically boosted immunization correlate with protection from mucosal SHIV challenge.Virology. 2008 Dec 20;382(2):217-25. doi: 10.1016/j.virol.2008.09.016. Epub 2008 Oct 22. Virology. 2008. PMID: 18947849 Free PMC article.
-
High-throughput profiling of anti-glycan humoral responses to SIV vaccination and challenge.PLoS One. 2013 Sep 23;8(9):e75302. doi: 10.1371/journal.pone.0075302. eCollection 2013. PLoS One. 2013. PMID: 24086502 Free PMC article.
References
-
- Amara, R. R., J. M. Smith, S. I. Staprans, D. C. Montefiori, F. Villinger, J. D. Altman, S. P. O'Neil, N. L. Kozyr, Y. Xu, L. S. Wyatt, P. L. Earl, J. G. Herndon, J. M. McNicholl, H. M. McClure, B. Moss, and H. L. Robinson. 2002. Critical role for Env as well as Gag-Pol in control of a simian-human immunodeficiency virus 89.6P challenge by a DNA prime/recombinant modified vaccinia virus Ankara vaccine. J. Virol. 76:6138-6146. - PMC - PubMed
-
- Baba, T. W., V. Liska, R. Hofmann-Lehmann, J. Vlasak, W. Xu, S. Ayehunie, L. A. Cavacini, M. R. Posner, H. Katinger, G. Stieglar, B. J. Barnacky, T. A. Rizvi, R. Schmidt, L. R. Hill, M. E. Keeling, J. Lu, J. E. Wright, T. C. Chou, and R. M. Ruprecht. 2000. Human neutralizing monoclonal antibodies of the IgG1 subtype protect against mucosal simian/human immunodeficiency virus infection. Nat. Med. 6:200-206. - PubMed
-
- Barker, T. D., D. Weissman, J. A. Daucher, K. M. Roche, and A. S. Fauci. 1996. Identification of multiple and distinct CD8+ T cell suppressor activities. J. Immunol. 156:4476-4483. - PubMed
-
- Barouch, D. H., S. Santra, J. E. Schmitz, M. J. Kuroda, T. M. Fu, W. Wagner, M. Bilska, A. Craiu, X. X. Zheng, G. R. Krivulka, K. Beaudry, M. A. Lifton, C. E. Nickerson, W. L. Trigona, K. Punt, D. C. Freed, L. Guan, S. Dubey, D. Casimiro, A. Simon, M. E. Davies, M. Chastain, T. B. Strom, R. S. Gelman, D. C. Montefiori, M. G. Lewis, E. A. Emini, J. W. Shiver, and N. L. Letvin. 2000. Control of viremia and prevention of clinical AIDS in rhesus monkeys by cytokine-augmented DNA vaccination. Science 290:486-492. - PubMed
-
- Bruce, C. B., A. Akrigg, S. A. Sharpe, T. Hanke, G. W. Wilkinson, and M. P. Cranage. 1999. Replication-deficient recombinant adenoviruses expressing the human immunodeficiency virus Env antigen can induce both humoral and CTL immune responses in mice. J. Gen. Virol. 80:2621-2628. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

