Alpha-galactosylceramide (KRN7000) suppression of chemical- and oncogene-dependent carcinogenesis
- PMID: 12867593
- PMCID: PMC170941
- DOI: 10.1073/pnas.1630663100
Alpha-galactosylceramide (KRN7000) suppression of chemical- and oncogene-dependent carcinogenesis
Abstract
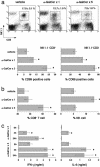
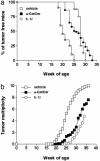
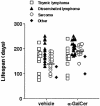
Recent studies have revealed significant efficacy of the marine sponge glycolipid, alpha-galactosylceramide (alpha-GalCer), in treatment of experimental metastatic cancers, infections, and autoimmune diseases. However, the capacity of alpha-GalCer to prevent tumor development had never, to our knowledge, been evaluated in mouse models of chemical- and oncogene-dependent carcinogenesis. In this study, we demonstrate that long-term administration of soluble alpha-GalCer, spanning the time of tumor initiation, inhibits primary tumor formation in three different models: methylcholanthrene-induced sarcomas, mammary carcinomas in Her-2/neu transgenic mice, and spontaneous sarcomas in p53-/- mice. Weekly treatment of mice with alpha-GalCer maintained lymphoid tissue natural killer cell and T cell activation and elevated serum IFN-gamma and IL-4 concentrations. Consistent with the antimetastatic activity of alpha-GalCer, prevention of methylcholanthrene-induced sarcoma was IFN-gammaand tumor necrosis factor-related apoptosis-inducing ligand dependent, but not perforin-dependent. Taken together, our results demonstrate that NK1.1+alphabetaTCR+ cell-based immune therapy can inhibit primary tumorigenesis.
Figures

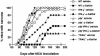
References
-
- Morita, M., Motoki, K., Akimoto, K., Natori, T., Sakai, T., Sawa, E., Yamaji, K., Koezuka, Y., Kobayashi, E. & Fukushima, H. (1995) J. Med. Chem. 38, 2176–2187. - PubMed
-
- Nakagawa, R., Motoki, K., Ueno, H., Iijima, R., Nakamura, H., Kobayashi, E., Shimosaka, A. & Koezuka, Y. (1998) Cancer Res. 58, 1202–1207. - PubMed
-
- Nakagawa, R., Serizawa, I., Motoki, K., Sato, M., Ueno, H., Iijima, R., Nakamura, H., Shimosaka, A. & Koezuka, Y. (2000) Oncol. Res. 12, 51–58. - PubMed
-
- Fuji, N., Ueda, Y., Fujiwara, H., Toh, T., Yoshimura, T. & Yamagishi, H. (2000) Clin. Cancer Res. 6, 3380–3387. - PubMed
-
- Kobayashi, E., Motoki, K., Uchida, T., Fukushima, H. & Koezuka, Y. (1995) Oncol. Res. 7, 529–534. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

