Cathepsin L in secretory vesicles functions as a prohormone-processing enzyme for production of the enkephalin peptide neurotransmitter
- PMID: 12869695
- PMCID: PMC170962
- DOI: 10.1073/pnas.1531542100
Cathepsin L in secretory vesicles functions as a prohormone-processing enzyme for production of the enkephalin peptide neurotransmitter
Abstract
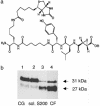
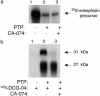
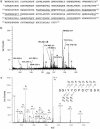
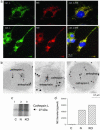
Multistep proteolytic mechanisms are essential for converting proprotein precursors into active peptide neurotransmitters and hormones. Cysteine proteases have been implicated in the processing of proenkephalin and other neuropeptide precursors. Although the papain family of cysteine proteases has been considered the primary proteases of the lysosomal degradation pathway, more recent studies indicate that functions of these enzymes are linked to specific biological processes. However, few protein substrates have been described for members of this family. We show here that secretory vesicle cathepsin L is the responsible cysteine protease of chromaffin granules for converting proenkephalin to the active enkephalin peptide neurotransmitter. The cysteine protease activity was identified as cathepsin L by affinity labeling with an activity-based probe for cysteine proteases followed by mass spectrometry for peptide sequencing. Production of [Met]enkephalin by cathepsin L occurred by proteolytic processing at dibasic and monobasic prohormone-processing sites. Cellular studies showed the colocalization of cathepsin L with [Met]enkephalin in secretory vesicles of neuroendocrine chromaffin cells by immunofluorescent confocal and immunoelectron microscopy. Functional localization of cathepsin L to the regulated secretory pathway was demonstrated by its cosecretion with [Met]enkephalin. Finally, in cathepsin L gene knockout mice, [Met]enkephalin levels in brain were reduced significantly; this occurred with an increase in the relative amounts of enkephalin precursor. These findings indicate a previously uncharacterized biological role for secretory vesicle cathepsin L in the production of [Met]enkephalin, an endogenous peptide neurotransmitter.
Figures

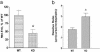
Similar articles
-
Evidence for functional localization of the proenkephalin-processing enzyme, prohormone thiol protease, to secretory vesicles of chromaffin cells.Endocrinology. 1999 Aug;140(8):3744-54. doi: 10.1210/endo.140.8.6926. Endocrinology. 1999. PMID: 10433235
-
Distinct properties of prohormone thiol protease (PTP) compared to cathepsins B, L, and H: evidence for PTP as a novel cysteine protease.Arch Biochem Biophys. 1994 Oct;314(1):171-7. doi: 10.1006/abbi.1994.1426. Arch Biochem Biophys. 1994. PMID: 7944391
-
Secretory vesicle aminopeptidase B related to neuropeptide processing: molecular identification and subcellular localization to enkephalin- and NPY-containing chromaffin granules.J Neurochem. 2007 Mar;100(5):1340-50. doi: 10.1111/j.1471-4159.2006.04325.x. Epub 2007 Jan 11. J Neurochem. 2007. PMID: 17241125
-
Cathepsin L and Arg/Lys aminopeptidase: a distinct prohormone processing pathway for the biosynthesis of peptide neurotransmitters and hormones.Biol Chem. 2004 Jun;385(6):473-80. doi: 10.1515/BC.2004.055. Biol Chem. 2004. PMID: 15255178 Review.
-
Cysteine Cathepsins in the secretory vesicle produce active peptides: Cathepsin L generates peptide neurotransmitters and cathepsin B produces beta-amyloid of Alzheimer's disease.Biochim Biophys Acta. 2012 Jan;1824(1):89-104. doi: 10.1016/j.bbapap.2011.08.015. Epub 2011 Sep 8. Biochim Biophys Acta. 2012. PMID: 21925292 Free PMC article. Review.
Cited by
-
Processing of capsid protein by cathepsin L plays a crucial role in replication of Japanese encephalitis virus in neural and macrophage cells.J Virol. 2007 Aug;81(16):8477-87. doi: 10.1128/JVI.00477-07. Epub 2007 Jun 6. J Virol. 2007. PMID: 17553875 Free PMC article.
-
Leukocyte telomere length and plasma catestatin and myeloid-related protein 8/14 concentrations in children with obstructive sleep apnea.Chest. 2010 Jul;138(1):91-9. doi: 10.1378/chest.09-2832. Epub 2010 Mar 18. Chest. 2010. PMID: 20299626 Free PMC article.
-
Proteomic analysis of cathepsin B- and L-deficient mouse brain lysosomes.Biochim Biophys Acta. 2007 Oct;1774(10):1237-46. doi: 10.1016/j.bbapap.2007.07.004. Epub 2007 Jul 19. Biochim Biophys Acta. 2007. PMID: 17765022 Free PMC article.
-
Impact of the Warhead of Dipeptidyl Keto Michael Acceptors on the Inhibition Mechanism of Cysteine Protease Cathepsin L.ACS Catal. 2023 Oct 3;13(20):13354-13368. doi: 10.1021/acscatal.3c02748. eCollection 2023 Oct 20. ACS Catal. 2023. PMID: 37881790 Free PMC article.
-
Toxoplasma gondii cathepsin L is the primary target of the invasion-inhibitory compound morpholinurea-leucyl-homophenyl-vinyl sulfone phenyl.J Biol Chem. 2009 Sep 25;284(39):26839-50. doi: 10.1074/jbc.M109.003780. Epub 2009 Jul 13. J Biol Chem. 2009. PMID: 19596863 Free PMC article.
References
-
- Gainer, H., Russell, J. T. & Loh, Y. P. (1985) Neuroendocrinology 40, 171-184. - PubMed
-
- Hook, V. Y. H., Azaryan, A. V. & Hwang, S. R. (1994) FASEB J. 8, 1269-1278. - PubMed
-
- Zhou, A., Webb, G., Zhu, X. R. & Steiner, D. F. (1999) J. Biol. Chem. 274, 20745-20748. - PubMed
-
- Seidah, N. G. & Prat, A. (2002) Essays Biochem. 38, 79-94. - PubMed
-
- Birch, N. P., Davie, A. D. & Christie, D. L. (1987) J. Biol. Chem. 262, 3382-3387. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

