Chemokine production and leukocyte recruitment to the lungs of Paracoccidioides brasiliensis-infected mice is modulated by interferon-gamma
- PMID: 12875978
- PMCID: PMC1868217
- DOI: 10.1016/s0002-9440(10)63686-3
Chemokine production and leukocyte recruitment to the lungs of Paracoccidioides brasiliensis-infected mice is modulated by interferon-gamma
Abstract
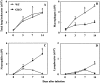
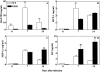
Chemokines and chemokine receptors play a role in cell recruitment during granulomatous inflammatory reactions. Here, we evaluated the expression of chemokines and chemokine receptors and their regulation by IFN-gamma in the course of Paracoccidioides brasiliensis (Pb) infection in mice. We found an association between KC and MIP-1alpha (CCL3) production and neutrophil infiltration in the lungs of Pb-infected mice during the early acute phase of infection. High levels of RANTES/CCL5, MCP-1/CCL2, IP-10/CXCL10, and Mig/CXCL9 simultaneously with mononuclear cell infiltration in the lungs was found. In the absence of IFN-gamma (GKO mice) we observed increased production of KC and MIP-1alpha and chronic neutrophilia. Moreover, we found a change in the chemokine receptor profiles expressed by wild-type (WT) versus GKO animals. Increased expression of CXCR3 and CCR5, and low levels of CCR3 and CCR4 were observed in the lungs of Pb-infected WT mice, whereas the opposite effect was observed in the lungs of GKO mice. Consistent with these results, infected cells from WT mice preferentially migrated in response to IP-10 (CXCR3 ligand), while those from GKO mice migrated in response to eotaxin/CCL11 (CCR3 ligand). These results suggest that IFN-gamma modulates the expression of chemokines and chemokine receptors as well as the kind of cells that infiltrate the lungs of Pb-infected mice.
Figures





Similar articles
-
Interferon-gamma and tumor necrosis factor-alpha determine resistance to Paracoccidioides brasiliensis infection in mice.Am J Pathol. 2000 May;156(5):1811-20. doi: 10.1016/s0002-9440(10)65053-5. Am J Pathol. 2000. PMID: 10793093 Free PMC article.
-
Chemokine expression during the development and resolution of a pulmonary leukocyte response to influenza A virus infection in mice.J Leukoc Biol. 2004 Oct;76(4):886-95. doi: 10.1189/jlb.1203644. Epub 2004 Jul 7. J Leukoc Biol. 2004. PMID: 15240757
-
Chemokine production during hypersensitivity pneumonitis.Eur J Immunol. 2004 Mar;34(3):677-685. doi: 10.1002/eji.200324634. Eur J Immunol. 2004. PMID: 14991597
-
The role of chemokines as inflammatory mediators in chronic hepatitis C virus infection.J Viral Hepat. 2007 Oct;14(10):675-87. doi: 10.1111/j.1365-2893.2006.00838.x. J Viral Hepat. 2007. PMID: 17875002 Review.
-
Hypersensitivity pneumonitis and alpha-chemokines.Clin Ter. 2017 Mar-Apr;168(2):e140-e145. doi: 10.7417/CT.2017.1996. Clin Ter. 2017. PMID: 28383627 Review.
Cited by
-
Pentoxifylline immunomodulation in the treatment of experimental chronic pulmonary paracoccidioidomycosis.Fibrogenesis Tissue Repair. 2015 Jun 1;8:10. doi: 10.1186/s13069-015-0027-8. eCollection 2015. Fibrogenesis Tissue Repair. 2015. PMID: 26034509 Free PMC article.
-
Proteomic Analysis of Paracoccidioides brasiliensis During Infection of Alveolar Macrophages Primed or Not by Interferon-Gamma.Front Microbiol. 2019 Feb 5;10:96. doi: 10.3389/fmicb.2019.00096. eCollection 2019. Front Microbiol. 2019. PMID: 30804901 Free PMC article.
-
Functional and phenotypic evaluation of eosinophils from patients with the acute form of paracoccidioidomycosis.PLoS Negl Trop Dis. 2017 May 10;11(5):e0005601. doi: 10.1371/journal.pntd.0005601. eCollection 2017 May. PLoS Negl Trop Dis. 2017. PMID: 28489854 Free PMC article.
-
CCR5 plays a critical role in the development of myocarditis and host protection in mice infected with Trypanosoma cruzi.J Infect Dis. 2005 Feb 15;191(4):627-36. doi: 10.1086/427515. Epub 2005 Jan 13. J Infect Dis. 2005. PMID: 15655788 Free PMC article.
-
Reconstituting NK Cells After Allogeneic Stem Cell Transplantation Show Impaired Response to the Fungal Pathogen Aspergillus fumigatus.Front Immunol. 2020 Sep 10;11:2117. doi: 10.3389/fimmu.2020.02117. eCollection 2020. Front Immunol. 2020. PMID: 33013893 Free PMC article.
References
-
- Restrepo A: The ecology of Paracoccidioides brasiliensis, a puzzle still unsolved. Sabouraudia 1985, 23:323-334 - PubMed
-
- Severo LC, Greyer GR, Londero AT, Porto NS, Rizzon CF: The primary pulmonary lymph node complex in paracoccidioidomycosis. Mycopathologia 1979, 67:115-118 - PubMed
-
- De Brito T, Franco MF: Granulomatous inflammation. Rev Inst Med Trop São Paulo 1994, 36:185-192 - PubMed
-
- Miyaji M, Nishimura K: Granuloma formation and killing functions of granuloma in congenitally athymic nude mice infected with Blastomyces dermatitides and Paracoccidioides brasiliensis. Mycopathologia 1983, 82:129-141 - PubMed
-
- Burger E, Vaz CCA, Sano A, Calich VLG, Singer-Vermes LM, Xidieh CF, Kashino SS, Nishimura K, Miyaji M: Paracoccidioides brasiliensis infection in nude mice: studies with isolates differing in virulence and definition of their T-cell-dependent and T-cell-independent components. Am J Trop Med Hyg 1996, 55:391-398 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

