Myeloid lineage switch of Pax5 mutant but not wild-type B cell progenitors by C/EBPalpha and GATA factors
- PMID: 12881423
- PMCID: PMC169053
- DOI: 10.1093/emboj/cdg380
Myeloid lineage switch of Pax5 mutant but not wild-type B cell progenitors by C/EBPalpha and GATA factors
Abstract
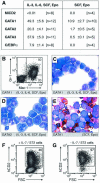
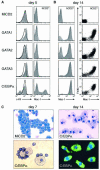
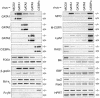
The developmental potential of hematopoietic progenitors is restricted early on to either the erythromyeloid or lymphoid lineages. The broad developmental potential of Pax5(-/-) pro-B cells is in apparent conflict with such a strict separation, although these progenitors realize the myeloid and erythroid potential with lower efficiency compared to the lymphoid cell fates. Here we demonstrate that ectopic expression of the transcription factors C/EBPalpha, GATA1, GATA2 and GATA3 strongly promoted in vitro macrophage differentiation and myeloid colony formation of Pax5(-/-) pro-B cells. GATA2 and GATA3 expression also resulted in efficient engraftment and myeloid development of Pax5(-/-) pro-B cells in vivo. The myeloid transdifferentiation of Pax5(-/-) pro-B cells was accompanied by the rapid activation of myeloid genes and concomitant repression of B-lymphoid genes by C/EBPalpha and GATA factors. These data identify the Pax5(-/-) pro-B cells as lymphoid progenitors with a latent myeloid potential that can be efficiently activated by myeloid transcription factors. The same regulators were unable to induce a myeloid lineage switch in Pax5(+/+) pro-B cells, indicating that Pax5 dominates over myeloid transcription factors in B-lymphocytes.
Figures



Similar articles
-
Antagonistic effect of CCAAT enhancer-binding protein-alpha and Pax5 in myeloid or lymphoid lineage choice in common lymphoid progenitors.Proc Natl Acad Sci U S A. 2006 Jan 17;103(3):672-7. doi: 10.1073/pnas.0510304103. Epub 2006 Jan 9. Proc Natl Acad Sci U S A. 2006. PMID: 16407117 Free PMC article.
-
Transcription factor EBF restricts alternative lineage options and promotes B cell fate commitment independently of Pax5.Nat Immunol. 2008 Feb;9(2):203-15. doi: 10.1038/ni1555. Epub 2008 Jan 6. Nat Immunol. 2008. PMID: 18176567
-
Analysis of Notch1 function by in vitro T cell differentiation of Pax5 mutant lymphoid progenitors.J Immunol. 2004 Sep 15;173(6):3935-44. doi: 10.4049/jimmunol.173.6.3935. J Immunol. 2004. PMID: 15356142
-
Fidelity and infidelity in commitment to B-lymphocyte lineage development.Immunol Rev. 2000 Jun;175:104-11. Immunol Rev. 2000. PMID: 10933595 Review.
-
Monoallelic expression of Pax5: a paradigm for the haploinsufficiency of mammalian Pax genes?Biol Chem. 1999 Jun;380(6):601-11. doi: 10.1515/BC.1999.077. Biol Chem. 1999. PMID: 10430025 Review.
Cited by
-
Comprehensive analysis of myeloid lineage conversion using mice expressing an inducible form of C/EBP alpha.EMBO J. 2006 Jul 26;25(14):3398-410. doi: 10.1038/sj.emboj.7601199. Epub 2006 Jul 6. EMBO J. 2006. PMID: 16858416 Free PMC article.
-
P2RY8-CRLF2Fusion-Positive Acute Myeloid Leukemia With Myelodysplasia-Related Changes: Response to Novel Therapy.JCO Precis Oncol. 2020;4:152-160. doi: 10.1200/PO.19.00294. Epub 2020 Mar 5. JCO Precis Oncol. 2020. PMID: 32395681 Free PMC article. No abstract available.
-
Next-Generation Sequencing in Acute Lymphoblastic Leukemia.Int J Mol Sci. 2019 Jun 15;20(12):2929. doi: 10.3390/ijms20122929. Int J Mol Sci. 2019. PMID: 31208040 Free PMC article. Review.
-
EP300-ZNF384 fusion gene product up-regulates GATA3 gene expression and induces hematopoietic stem cell gene expression signature in B-cell precursor acute lymphoblastic leukemia cells.Int J Hematol. 2017 Aug;106(2):269-281. doi: 10.1007/s12185-017-2220-6. Epub 2017 Apr 4. Int J Hematol. 2017. PMID: 28378055
-
The lymphoid lineage-specific actin-uncapping protein Rltpr is essential for costimulation via CD28 and the development of regulatory T cells.Nat Immunol. 2013 Aug;14(8):858-66. doi: 10.1038/ni.2634. Epub 2013 Jun 23. Nat Immunol. 2013. PMID: 23793062
References
-
- Akashi K., Traver,D., Miyamoto,T. and Weissman,I.L. (2000) A clonogenic common myeloid progenitor that gives rise to all myeloid lineages. Nature, 404, 193–197. - PubMed
-
- Allman D., Sambandam,A., Kim,S., Miller,J.P., Pagan,A., Well,D., Meraz,A. and Bhandoola,A. (2003) Thymopoiesis independent of common lymphoid progenitors. Nat. Immunol., 4, 168–174. - PubMed
-
- Andrews N.C. and Orkin,S.H. (1994) Transcriptional control of erythropoiesis. Curr. Opin. Hematol., 1, 119–124. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

