Essential role of RelA Ser311 phosphorylation by zetaPKC in NF-kappaB transcriptional activation
- PMID: 12881425
- PMCID: PMC169043
- DOI: 10.1093/emboj/cdg370
Essential role of RelA Ser311 phosphorylation by zetaPKC in NF-kappaB transcriptional activation
Abstract
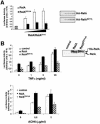
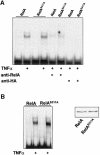
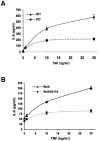
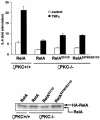
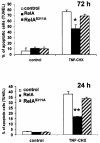
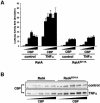
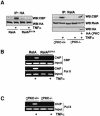
The activation of the transcription factor NF-kappaB is central to the control of the cellular response triggered by many stimuli. Once released from the inhibitory molecule IkappaB, NF-kappaB is translocated to the nucleus, and it has to be phosphorylated to activate transcription. In zeta protein kinase C (PKC)-deficient cells, NF-kappaB is transcriptionally inactive and the phosphorylation of the RelA subunit in response to tumor necrosis factor (TNF-alpha) is severely impaired. In vitro assays showed that zetaPKC directly phosphorylates RelA. Here we demonstrate that Ser311 accounts for zetaPKC phosphorylation of RelA and that this site is phosphorylated in vivo in response to TNF-alpha. Also, an inactivating mutation of that residue severely impairs RelA transcriptional activity, blocks its anti-apoptotic function and abrogates the interaction of RelA with the co-activator CBP as well as its recruitment, and that of RNA polymerase II (Pol II) with the interleukin-6 (IL-6) promoter. The interaction of endogenous CBP with endogenous RelA is inhibited in zetaPKC-/- cells, as well as the binding of Pol II to the IL-6 promoter. These results demonstrate the mechanism whereby zetaPKC regulates NF-kappaB activation in vivo.
Figures


References
-
- Anrather J., Csizmadia,V., Soares,M.P. and Winkler,H. (1999) Regulation of NF-κB RelA phosphorylation and transcriptional activity by p21(ras) and protein kinase Cζ in primary endothelial cells. J. Biol. Chem., 274, 13594–13603. - PubMed
-
- Claudio E., Brown,K., Park,S., Wang,H. and Siebenlist,U. (2002) BAFF-induced NEMO-independent processing of NF-κB2 in maturing B cells. Nat. Immunol., 3, 958–965. - PubMed
-
- Cowley S., Paterson,H., Kemp,P. and Marshall,C.J. (1994) Activation of MAP kinase kinase is necessary and sufficient for PC12 differentiation and for transformation of NIH 3T3 cells. Cell, 77, 841–852. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

