Side-chain charge effects and conductance determinants in the pore of ClC-0 chloride channels
- PMID: 12885875
- PMCID: PMC2229543
- DOI: 10.1085/jgp.200308844
Side-chain charge effects and conductance determinants in the pore of ClC-0 chloride channels
Abstract
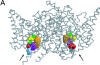
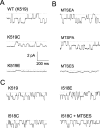
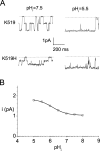
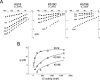
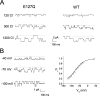
The charge on the side chain of the internal pore residue lysine 519 (K519) of the Torpedo ClC-0 chloride (Cl-) channel affects channel conductance. Experiments that replace wild-type (WT) lysine with neutral or negatively charged residues or that modify the K519C mutant with various methane thiosulfonate (MTS) reagents show that the conductance of the channel decreases when the charge at position 519 is made more negative. This charge effect on the channel conductance diminishes in the presence of a high intracellular Cl- concentration ([Cl-]i). However, the application of high concentrations of nonpermeant ions, such as glutamate or sulfate (SO42-), does not change the conductance, suggesting that the electrostatic effects created by the charge at position 519 are unlikely due to a surface charge mechanism. Another pore residue, glutamate 127 (E127), plays an even more critical role in controlling channel conductance. This negatively charged residue, based on the structures of the homologous bacterial ClC channels, lies 4-5 A from K519. Altering the charge of this residue can influence the apparent Cl- affinity as well as the saturated pore conductance in the conductance-Cl- activity curve. Amino acid residues at the selectivity filter also control the pore conductance but mutating these residues mainly affects the maximal pore conductance. These results suggest at least two different conductance determinants in the pore of ClC-0, consistent with the most recent crystal structure of the bacterial ClC channel solved to 2.5 A, in which multiple Cl--binding sites were identified in the pore. Thus, we suggest that the occupancy of the internal Cl--binding site is directly controlled by the charged residues located at the inner pore mouth. On the other hand, the Cl--binding site at the selectivity filter controls the exit rate of Cl- and therefore determines the maximal channel conductance.
Figures






Comment in
-
ClC channels: reading eukaryotic function through prokaryotic spectacles.J Gen Physiol. 2003 Aug;122(2):129-31. doi: 10.1085/jgp.200308898. J Gen Physiol. 2003. PMID: 12885874 Free PMC article. No abstract available.
References
-
- Andersen, O.S. 1989. Kinetics of ion movement mediated by carriers and channels. Methods Enzymol. 171:62–112. - PubMed

