Endocytosis of hepatitis B immune globulin into hepatocytes inhibits the secretion of hepatitis B virus surface antigen and virions
- PMID: 12885906
- PMCID: PMC167249
- DOI: 10.1128/jvi.77.16.8882-8892.2003
Endocytosis of hepatitis B immune globulin into hepatocytes inhibits the secretion of hepatitis B virus surface antigen and virions
Abstract
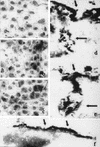
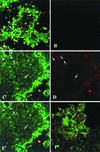
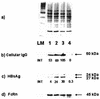
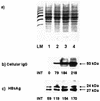
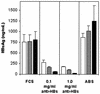
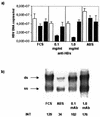
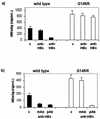
Hepatitis B immunoglobulin is used for prophylaxis against hepatitis B virus (HBV) and is thought to act by neutralization of virions and hepatitis B virus surface antigen (HBsAg)-containing particles in circulation. Using a panel of hepatocyte-derived cell lines, the present study investigated in vitro whether HBs-specific immunoglobulin G (IgG) is internalized in hepatocytes and whether it interacts with HBsAg in the cells. By immunoelectron microscopy and immunoblotting, human IgG and FcRn receptor for IgG were demonstrated on cellular membranes and in cytoplasmic extracts, irrespective of the HBsAg status of the cells. Furthermore, HBsAg and anti-HBs were shown to be colocalized in the same cellular compartment by two-color confocal microscopy. Endocytosis of HBs-specific IgG caused intracellular accumulation of HBsAg in a dose-dependent manner and inhibited the secretion of HBsAg and HBV virions from the cells. These effects were not observed with F(ab)(2) fragments or nonimmune IgG as controls. The specificity of intracellular HBsAg- anti-HBs interaction was further investigated in cells transfected with HBV genomes expressing wild-type HBsAg or immune escape HBsAg (with a G145R mutation). Monoclonal anti-HBs markedly reduced the secretion of wild-type HBsAg, while the secretion of mutant HBsAg was not affected. These results suggest that HBs-specific IgG binds to hepatocytes and interacts with HBsAg within the cells. This may be relevant for the selection of surface antibody escape mutations.
Figures

Similar articles
-
De novo activation of HBV with escape mutations from hepatitis B surface antibody after living donor liver transplantation.Antivir Ther. 2011;16(4):479-87. doi: 10.3851/IMP1771. Antivir Ther. 2011. PMID: 21685535
-
Coexistence of hepatitis B surface antigen (HBsAg) and heterologous subtype-specific antibodies to HBsAg among patients with chronic hepatitis B virus infection.Clin Infect Dis. 2007 May 1;44(9):1161-9. doi: 10.1086/513200. Epub 2007 Mar 19. Clin Infect Dis. 2007. PMID: 17407033
-
N-glycosylation mutations within hepatitis B virus surface major hydrophilic region contribute mostly to immune escape.J Hepatol. 2014 Mar;60(3):515-22. doi: 10.1016/j.jhep.2013.11.004. Epub 2013 Nov 13. J Hepatol. 2014. PMID: 24239777
-
Multiple epitopes of hepatitis B virus surface antigen targeted by human plasma-derived immunoglobulins coincide with clinically observed escape mutations.J Med Virol. 2022 Feb;94(2):649-658. doi: 10.1002/jmv.27278. Epub 2021 Aug 26. J Med Virol. 2022. PMID: 34406663 Free PMC article.
-
An Overview of Hepatitis B Virus Surface Antigen Secretion Inhibitors.Front Microbiol. 2018 Apr 5;9:662. doi: 10.3389/fmicb.2018.00662. eCollection 2018. Front Microbiol. 2018. PMID: 29675010 Free PMC article. Review.
Cited by
-
Prevention of HBV Recurrence after Liver Transplant: A Review.J Clin Transl Hepatol. 2020 Jun 28;8(2):150-160. doi: 10.14218/JCTH.2020.00003. Epub 2020 May 25. J Clin Transl Hepatol. 2020. PMID: 32832395 Free PMC article. Review.
-
Unusual naturally occurring humoral and cellular mutated epitopes of hepatitis B virus in a chronically infected argentine patient with anti-HBs antibodies.J Clin Microbiol. 2006 Jun;44(6):2191-8. doi: 10.1128/JCM.00057-06. J Clin Microbiol. 2006. PMID: 16757620 Free PMC article.
-
A bispecific antibody against two different epitopes on hepatitis B surface antigen has potent hepatitis B virus neutralizing activity.MAbs. 2013 Nov-Dec;5(6):946-55. doi: 10.4161/mabs.26390. MAbs. 2013. PMID: 24492346 Free PMC article.
-
Contradictory immune response in post liver transplantation hepatitis B and C.Int J Inflam. 2014;2014:814760. doi: 10.1155/2014/814760. Epub 2014 Aug 24. Int J Inflam. 2014. PMID: 25215259 Free PMC article. Review.
-
Fc gamma receptor-like hepatitis C virus core protein binds differentially to IgG of discordant Fc (GM) genotypes.Mol Immunol. 2007 Jul;44(15):3805-8. doi: 10.1016/j.molimm.2007.03.022. Epub 2007 May 7. Mol Immunol. 2007. PMID: 17485114 Free PMC article.
References
-
- Carman, W. F., A. R. Zanetti, P. Karayiannis, J. Waters, G. Manzillo, E. Tanzi, A. J. Zuckerman, and H. C. Thomas. 1990. Vaccine-induced escape mutant of hepatitis B virus. Lancet 336:325-329. - PubMed
-
- Carman, W. F., C. Trautwein, F. J. van Deursen, K. Colman, E. Dornan, G. McIntyre, J. Walters, V. Kliem, R. Muller, H. C. Thomas, and M. P. Manns. 1996. Hepatitis B envelope variation after transplantation with and without hepatitis B immune globulin prophylaxis. Hepatology 24:489-493. - PubMed
-
- Carman, W. F., A. Owsianka, L. A. Wallace, B. C. Dow, and D. J. Mutimer. 1999. Antigenic characterisation of pre- and post-liver transplant hepatitis B surface antigen sequences from patients treated with hepatitis B immune globulin. J. Hepatol. 31:195-201. - PubMed
-
- Cooreman, M. P., M. H. van Roosmalen, R. te Morsche, C. M. Sunnen, E. M. de Ven, J. B. Jansen, G. N. Tytgat, P. L. de Wit, and W. P. Paulij. 1999. Characterization of the reactivity pattern of murine antibodies against wild-type hepatitis B surface antigen to G145R and other naturally occurring “a” loop escape mutations. Hepatology 30:1287-1292. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

