Mechanisms of P/CAF auto-acetylation
- PMID: 12888487
- PMCID: PMC169960
- DOI: 10.1093/nar/gkg655
Mechanisms of P/CAF auto-acetylation
Abstract
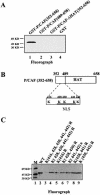
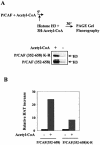
P/CAF is a histone acetyltransferase enzyme which was originally identified as a CBP/p300-binding protein. In this manuscript we report that human P/CAF is acetylated in vivo. We find that P/CAF is acetylated by itself and by p300 but not by CBP. P/CAF acetylation can be an intra- or intermolecular event. The intermolecular acetylation requires the N-terminal domain of P/CAF. The intramolecular acetylation targets five lysines (416-442) at the P/CAF C-terminus, which are in the nuclear localisation signal (NLS). Finally, we show that acetylation of P/CAF leads to an increment of its histone acetyltransferase (HAT) activity. These findings identify a new post-translation modification on P/CAF which may regulate its function.
Figures






References
-
- Konberg R.D. and Lorch,Y. (1999) Chromatin-modifying and remodelling complexes. Curr. Opin. Genet. Dev., 9, 148–151. - PubMed
-
- Kingston R.E. and Narlikar,G.J. (1999) ATP-dependent remodelling and acetylation as regulators of chromatin fluidity. Genes Dev., 13, 2339–2352. - PubMed
-
- Wu J. and Grunstein,M. (2000) 25 years after the nucleosome model: chromatin modifications. Trends Biochem. Sci., 25, 619–623. - PubMed
-
- Berger S. (2002) Histone modifications in transcriptional regulation. Curr. Opin. Genet. Dev., 12, 142–148. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

