Critical amino acids in the active site of meprin metalloproteinases for substrate and peptide bond specificity
- PMID: 12888571
- PMCID: PMC2366086
- DOI: 10.1074/jbc.M303718200
Critical amino acids in the active site of meprin metalloproteinases for substrate and peptide bond specificity
Abstract
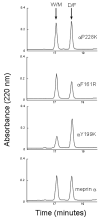
The protease domains of the evolutionarily related alpha and beta subunits of meprin metalloproteases are approximately 55% identical at the amino acid level; however, their substrate and peptide bond specificities differ markedly. The meprin beta subunit favors acidic residues proximal to the scissile bond, while the alpha subunit prefers small or aromatic amino acids flanking the scissile bond. Thus gastrin, a peptide that contains a string of five Glu residues, is an excellent substrate for meprin beta, while it is not hydrolyzed by meprin alpha. Work herein aimed to identify critical amino acids in the meprin active sites that determine the substrate specificity differences. Sequence alignments and homology models, based on the crystal structure of the crayfish astacin, showed electrostatic differences within the meprin active sites. Site-directed mutagenesis of active site residues demonstrated that replacement of a hydrophobic residue by a basic amino acid enabled the meprin alpha protease to cleave gastrin. The meprin alphaY199K mutant was most effective; the corresponding mutation of meprin betaK185Y resulted in decreased activity toward gastrin. Peptide cleavage site determinations and kinetic analyses using a variety of peptides extended evidence that meprin alphaTyr-199/betaLys-185 are substrate specificity determinants in meprin active sites. These studies shed light on the molecular basis for the substrate specificity differences of astacin metalloproteinases.
Figures


Similar articles
-
Marked differences between metalloproteases meprin A and B in substrate and peptide bond specificity.J Biol Chem. 2001 Apr 20;276(16):13248-55. doi: 10.1074/jbc.M011414200. Epub 2001 Jan 22. J Biol Chem. 2001. PMID: 11278902
-
Unveiling the substrate specificity of meprin beta on the basis of the site in protein kinase A cleaved by the kinase splitting membranal proteinase.J Biol Chem. 1997 Feb 7;272(6):3153-60. doi: 10.1074/jbc.272.6.3153. J Biol Chem. 1997. PMID: 9013548
-
Proteomic analyses reveal an acidic prime side specificity for the astacin metalloprotease family reflected by physiological substrates.Mol Cell Proteomics. 2011 Sep;10(9):M111.009233. doi: 10.1074/mcp.M111.009233. Epub 2011 Jun 21. Mol Cell Proteomics. 2011. PMID: 21693781 Free PMC article.
-
Meprin metalloproteases: Molecular regulation and function in inflammation and fibrosis.Biochim Biophys Acta Mol Cell Res. 2017 Nov;1864(11 Pt B):2096-2104. doi: 10.1016/j.bbamcr.2017.05.011. Epub 2017 May 11. Biochim Biophys Acta Mol Cell Res. 2017. PMID: 28502593 Review.
-
The metalloproteases meprin α and meprin β: unique enzymes in inflammation, neurodegeneration, cancer and fibrosis.Biochem J. 2013 Mar 1;450(2):253-64. doi: 10.1042/BJ20121751. Biochem J. 2013. PMID: 23410038 Free PMC article. Review.
Cited by
-
Engineering of TEV protease variants by yeast ER sequestration screening (YESS) of combinatorial libraries.Proc Natl Acad Sci U S A. 2013 Apr 30;110(18):7229-34. doi: 10.1073/pnas.1215994110. Epub 2013 Apr 15. Proc Natl Acad Sci U S A. 2013. PMID: 23589865 Free PMC article.
-
Synthesis and structure-activity relationships of pyrazole-based inhibitors of meprin α and β.J Enzyme Inhib Med Chem. 2023 Dec;38(1):2165648. doi: 10.1080/14756366.2023.2165648. J Enzyme Inhib Med Chem. 2023. PMID: 36661029 Free PMC article.
-
Structure and Dynamics of Meprin β in Complex with a Hydroxamate-Based Inhibitor.Int J Mol Sci. 2021 May 26;22(11):5651. doi: 10.3390/ijms22115651. Int J Mol Sci. 2021. PMID: 34073350 Free PMC article.
-
Engineering next generation proteases.Curr Opin Biotechnol. 2009 Aug;20(4):390-7. doi: 10.1016/j.copbio.2009.07.003. Epub 2009 Aug 24. Curr Opin Biotechnol. 2009. PMID: 19709875 Free PMC article. Review.
-
Villin and actin in the mouse kidney brush-border membrane bind to and are degraded by meprins, an interaction that contributes to injury in ischemia-reperfusion.Am J Physiol Renal Physiol. 2011 Oct;301(4):F871-82. doi: 10.1152/ajprenal.00703.2010. Epub 2011 Jul 27. Am J Physiol Renal Physiol. 2011. PMID: 21795642 Free PMC article.
References
-
- Bertenshaw GP, Turk BE, Hubbard SJ, Matters GL, Bylander JE, Crisman JM, Cantley LC, Bond JS. J Biol Chem. 2001;276:13248–13255. - PubMed
-
- Johnson GD, Bond JS. In: Handbook of Proteolytic Enzymes. Barrett AJ, Woessner F, Rawlings N, editors. Academic Press, Inc; San Diego, CA: 1998. pp. 1222–1229.
-
- Bertenshaw GP, Norcum MT, Bond JS. J Biol Chem. 2003;278:2522–2532. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

