Role of Pseudomonas putida tol-oprL gene products in uptake of solutes through the cytoplasmic membrane
- PMID: 12896989
- PMCID: PMC166457
- DOI: 10.1128/JB.185.16.4707-4716.2003
Role of Pseudomonas putida tol-oprL gene products in uptake of solutes through the cytoplasmic membrane
Abstract
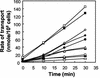
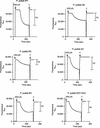
Proteins of the Tol-Pal (Tol-OprL) system play a key role in the maintenance of outer membrane integrity and cell morphology in gram-negative bacteria. Here we describe an additional role for this system in the transport of various carbon sources across the cytoplasmic membrane. Growth of Pseudomonas putida tol-oprL mutant strains in minimal medium with glycerol, fructose, or arginine was impaired, and the growth rate with succinate, proline, or sucrose as the carbon source was lower than the growth rate of the parental strain. Assays with radiolabeled substrates revealed that the rates of uptake of these compounds by mutant cells were lower than the rates of uptake by the wild-type strain. The pattern and amount of outer membrane protein in the P. putida tol-oprL mutants were not changed, suggesting that the transport defect was not in the outer membrane. Consistently, the uptake of radiolabeled glucose and glycerol in spheroplasts was defective in the P. putida tol-oprL mutant strains, suggesting that there was a defect at the cytoplasmic membrane level. Generation of a proton motive force appeared to be unaffected in these mutants. To rule out the possibility that the uptake defect was due to a lack of specific transporter proteins, the PutP symporter was overproduced, but this overproduction did not enhance proline uptake in the tol-oprL mutants. These results suggest that the Tol-OprL system is necessary for appropriate functioning of certain uptake systems at the level of the cytoplasmic membrane.
Figures

Similar articles
-
Transcriptional organization of the Pseudomonas putida tol-oprL genes.J Bacteriol. 2003 Jan;185(1):184-95. doi: 10.1128/JB.185.1.184-195.2003. J Bacteriol. 2003. PMID: 12486055 Free PMC article.
-
RegA, iron, and growth phase regulate expression of the Pseudomonas aeruginosa tol-oprL gene cluster.J Bacteriol. 2000 Apr;182(8):2077-87. doi: 10.1128/JB.182.8.2077-2087.2000. J Bacteriol. 2000. PMID: 10735848 Free PMC article.
-
Characterization of an OprL null mutant of Pseudomonas putida.J Bacteriol. 1996 Oct;178(19):5836-40. doi: 10.1128/jb.178.19.5836-5840.1996. J Bacteriol. 1996. PMID: 8824639 Free PMC article.
-
The Tol-Pal proteins of the Escherichia coli cell envelope: an energized system required for outer membrane integrity?Res Microbiol. 2001 Jul-Aug;152(6):523-9. doi: 10.1016/s0923-2508(01)01226-8. Res Microbiol. 2001. PMID: 11501670 Review.
-
The Tol proteins of Escherichia coli and their involvement in the uptake of biomolecules and outer membrane stability.FEMS Microbiol Lett. 1999 Aug 15;177(2):191-7. doi: 10.1111/j.1574-6968.1999.tb13731.x. FEMS Microbiol Lett. 1999. PMID: 10474183 Review.
Cited by
-
Lipoprotein biosynthesis as a target for anti-Wolbachia treatment of filarial nematodes.Parasit Vectors. 2010 Oct 14;3:99. doi: 10.1186/1756-3305-3-99. Parasit Vectors. 2010. PMID: 20946650 Free PMC article.
-
Comprehensive analyses of transport proteins encoded within the genome of "Aromatoleum aromaticum" strain EbN1.J Membr Biol. 2009 May;229(2):53-90. doi: 10.1007/s00232-009-9168-2. Epub 2009 Jun 9. J Membr Biol. 2009. PMID: 19506936
-
Polar localization of Escherichia coli chemoreceptors requires an intact Tol-Pal complex.Mol Microbiol. 2014 Jun;92(5):985-1004. doi: 10.1111/mmi.12609. Epub 2014 May 2. Mol Microbiol. 2014. PMID: 24720726 Free PMC article.
-
Functional analysis of aromatic biosynthetic pathways in Pseudomonas putida KT2440.Microb Biotechnol. 2009 Jan;2(1):91-100. doi: 10.1111/j.1751-7915.2008.00062.x. Epub 2008 Oct 13. Microb Biotechnol. 2009. PMID: 21261884 Free PMC article.
-
Defective O-antigen polymerization in tolA and pal mutants of Escherichia coli in response to extracytoplasmic stress.J Bacteriol. 2005 May;187(10):3359-68. doi: 10.1128/JB.187.10.3359-3368.2005. J Bacteriol. 2005. PMID: 15866920 Free PMC article.
References
-
- Adewoye, L. O., L. Tschetter, J. O'Neil, and E. A. Worobec. 1998. Channel specificity and secondary structure of the glucose-inducible porins of Pseudomonas sp. J. Bioenerg. Biomembr. 30:257-267. - PubMed
-
- Adewoye, L. O., and E. A. Worobec. 2000. Identification and characterization of the gltK gene encoding a membrane-associated glucose transport protein of Pseudomonas aeruginosa. Gene 253:323-330. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

