Mouse mammary tumor virus c-rel transgenic mice develop mammary tumors
- PMID: 12897145
- PMCID: PMC166341
- DOI: 10.1128/MCB.23.16.5738-5754.2003
Mouse mammary tumor virus c-rel transgenic mice develop mammary tumors
Abstract
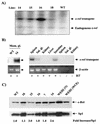
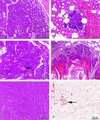
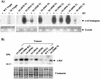
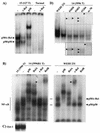
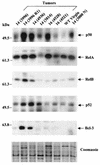
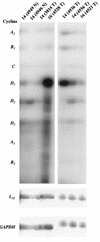
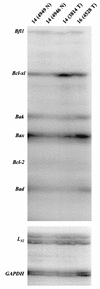
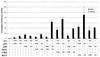
Amplification, overexpression, or rearrangement of the c-rel gene, encoding the c-Rel NF-kappaB subunit, has been reported in solid and hematopoietic malignancies. For example, many primary human breast cancer tissue samples express high levels of nuclear c-Rel. While the Rev-T oncogene v-rel causes tumors in birds, the ability of c-Rel to transform in vivo has not been demonstrated. To directly test the role of c-Rel in breast tumorigenesis, mice were generated in which overexpression of mouse c-rel cDNA was driven by the hormone-responsive mouse mammary tumor virus long terminal repeat (MMTV-LTR) promoter, and four founder lines identified. In the first cycle of pregnancy, the expression of transgenic c-rel mRNA was observed, and levels of c-Rel protein were increased in the mammary gland. Importantly, 31.6% of mice developed one or more mammary tumors at an average age of 19.9 months. Mammary tumors were of diverse histology and expressed increased levels of nuclear NF-kappaB. Analysis of the composition of NF-kappaB complexes in the tumors revealed aberrant nuclear expression of multiple subunits, including c-Rel, p50, p52, RelA, RelB, and the Bcl-3 protein, as observed previously in human primary breast cancers. Expression of the cancer-related NF-kappaB target genes cyclin D1, c-myc, and bcl-xl was significantly increased in grossly normal transgenic mammary glands starting the first cycle of pregnancy and increased further in mammary carcinomas compared to mammary glands from wild-type mice or virgin transgenic mice. In transient transfection analysis in untransformed breast epithelial cells, c-Rel-p52 or -p50 heterodimers either potently or modestly induced cyclin D1 promoter activity, respectively. Lastly, stable overexpression of c-Rel resulted in increased cyclin D1 and NF-kappaB p52 and p50 subunit protein levels. These results indicate for the first time that dysregulated expression of c-Rel, as observed in breast cancers, is capable of contributing to mammary tumorigenesis.
Figures




References
-
- Abbadie, C., N. Kabrun, F. Bouali, J. Smardova, D. Stehelin, B. Vandenbunder, and P. J. Enrietto. 1993. High levels of c-rel expression are associated with programmed cell death in the developing avian embryo and in bone marrow cells in vitro. Cell 75:899-912. - PubMed
-
- Alizadeh, A. A., M. B. Eisen, R. E. Davis, C. Ma, I. S. Lossos, A. Rosenwald, J. C. Boldrick, H. Sabet, T. Tran, X. Yu, J. I. Powell, L. Yang, G. E. Marti, T. Moore, J. Hudson, Jr., L. Lu, D. B. Lewis, R. Tibshirani, G. Sherlock, W. C. Chan, T. C. Greiner, D. D. Weisenburger, J. O. Armitage, R. Warnke, L. M. Staudt, and et al. 2000. Distinct types of diffuse large B-cell lymphoma identified by gene expression profiling. Nature 403:503-511. - PubMed
-
- Arsura, M., M. J. FitzGerald, N. Fausto, and G. E. Sonenshein. 1997. Nuclear factor-κB/Rel blocks transforming growth factor beta1-induced apoptosis of murine hepatocyte cell lines. Cell Growth Differ. 8:1049-1059. - PubMed
-
- Arsura, M., and G. E. Sonenshein. 2001. The roles of the c-myc and c-myb oncogenes in hematopoiesis and leukemogenesis, p. 521-549. In K. Ravid and J. Licht (ed.), Transcription factors: normal and malignant development of blood cells. Wiley-Liss Inc., New York, N.Y.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
