Replication checkpoint kinase Cds1 regulates recombinational repair protein Rad60
- PMID: 12897162
- PMCID: PMC166335
- DOI: 10.1128/MCB.23.16.5939-5946.2003
Replication checkpoint kinase Cds1 regulates recombinational repair protein Rad60
Abstract
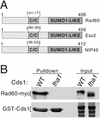
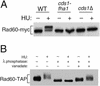
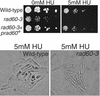
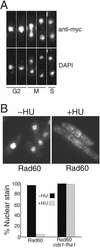
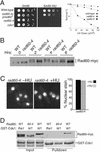
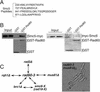
Genome integrity is protected by Cds1 (Chk2), a checkpoint kinase that stabilizes arrested replication forks. How Cds1 accomplishes this task is unknown. We report that Cds1 interacts with Rad60, a protein required for recombinational repair in fission yeast. Cds1 activation triggers Rad60 phosphorylation and nuclear delocalization. A Rad60 mutant that inhibits regulation by Cds1 renders cells specifically sensitive to replication fork arrest. Genetic and biochemical studies indicate that Rad60 functions codependently with Smc5 and Smc6, subunits of an SMC (structural maintenance of chromosomes) complex required for recombinational repair. These studies indicate that regulation of Rad60 is an important part of the replication checkpoint response controlled by Cds1. We propose that control of Rad60 regulates recombination events at stalled forks.
Figures
References
-
- Aono, N., T. Sutani, T. Tomonaga, S. Mochida, and M. Yanagida. 2002. Cnd2 has dual roles in mitotic condensation and interphase. Nature 417:197-202. - PubMed
-
- Birkenbihl, R. P., and S. Subramani. 1995. The rad21 gene product of Schizosaccharomyces pombe is a nuclear, cell cycle-regulated phosphoprotein. J. Biol. Chem. 270:7703-7711. - PubMed
-
- Boddy, M. N., B. Furnari, O. Mondesert, and P. Russell. 1998. Replication checkpoint enforced by kinases Cds1 and Chk1. Science 280:909-912. - PubMed
-
- Boddy, M. N., P. H. Gaillard, W. H. McDonald, P. Shanahan, J. R. Yates III, and P. Russell. 2001. Mus81-Eme1 are essential components of a Holliday junction resolvase. Cell 107:537-548. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
