Dissociation of amyloid fibrils of alpha-synuclein and transthyretin by pressure reveals their reversible nature and the formation of water-excluded cavities
- PMID: 12900507
- PMCID: PMC187855
- DOI: 10.1073/pnas.1734009100
Dissociation of amyloid fibrils of alpha-synuclein and transthyretin by pressure reveals their reversible nature and the formation of water-excluded cavities
Abstract
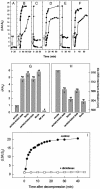
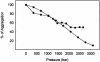
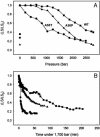
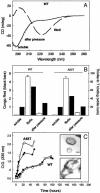
Protein misfolding and aggregation have been linked to several human diseases, including Alzheimer's disease, Parkinson's disease, and systemic amyloidosis, by mechanisms that are not yet completely understood. The hallmark of most of these diseases is the formation of highly ordered and beta-sheet-rich aggregates referred to as amyloid fibrils. Fibril formation by WT transthyretin (TTR) or TTR variants has been linked to the etiology of systemic amyloidosis and familial amyloid polyneuropathy, respectively. Similarly, amyloid fibril formation by alpha-synuclein (alpha-syn) has been linked to neurodegeneration in Parkinson's disease, a movement disorder characterized by selective degeneration of dopaminergic neurons in the substantia nigra. Here we show that consecutive cycles of compression-decompression under aggregating conditions lead to reversible dissociation of TTR and alpha-syn fibrils. The high sensitivity of amyloid fibrils toward high hydrostatic pressure (HHP) indicates the existence of packing defects in the fibril core. In addition, through the use of HHP we are able to detect differences in stability between fibrils formed from WT TTR and the familial amyloidotic polyneuropathy-associated variant V30M. The fibrils formed by WT alpha-syn were less susceptible to pressure denaturation than the Parkinson's disease-linked variants, A30P and A53T. This finding implies that fibrils of alpha-syn formed from the variants would be more easily dissolved into small oligomers by the cellular machinery. This result has physiological importance in light of the current view that the pathogenic species are the small aggregates rather the mature fibrils. Finally, the HHP-induced formation of fibrils from TTR is relatively fast (approximately 60 min), a quality that allows screening of antiamyloidogenic drugs.
Figures
Similar articles
-
Hydration and packing are crucial to amyloidogenesis as revealed by pressure studies on transthyretin variants that either protect or worsen amyloid disease.J Mol Biol. 2003 May 9;328(4):963-74. doi: 10.1016/s0022-2836(03)00368-1. J Mol Biol. 2003. PMID: 12729768
-
Pressure effects on α-synuclein amyloid fibrils: An experimental investigation on their dissociation and reversible nature.Arch Biochem Biophys. 2017 Aug 1;627:46-55. doi: 10.1016/j.abb.2017.06.007. Epub 2017 Jun 15. Arch Biochem Biophys. 2017. PMID: 28624352
-
Fibrils formed in vitro from alpha-synuclein and two mutant forms linked to Parkinson's disease are typical amyloid.Biochemistry. 2000 Mar 14;39(10):2552-63. doi: 10.1021/bi991447r. Biochemistry. 2000. PMID: 10704204
-
Protein denaturation and aggregation: Cellular responses to denatured and aggregated proteins.Ann N Y Acad Sci. 2005 Dec;1066:181-221. doi: 10.1196/annals.1363.030. Ann N Y Acad Sci. 2005. PMID: 16533927 Review.
-
Pressure-temperature folding landscape in proteins involved in neurodegenerative diseases and cancer.Biophys Chem. 2013 Dec 15;183:9-18. doi: 10.1016/j.bpc.2013.06.002. Epub 2013 Jun 14. Biophys Chem. 2013. PMID: 23849959 Review.
Cited by
-
Cavities determine the pressure unfolding of proteins.Proc Natl Acad Sci U S A. 2012 May 1;109(18):6945-50. doi: 10.1073/pnas.1200915109. Epub 2012 Apr 10. Proc Natl Acad Sci U S A. 2012. PMID: 22496593 Free PMC article.
-
Ligand binding and hydration in protein misfolding: insights from studies of prion and p53 tumor suppressor proteins.Acc Chem Res. 2010 Feb 16;43(2):271-9. doi: 10.1021/ar900179t. Acc Chem Res. 2010. PMID: 19817406 Free PMC article. Review.
-
High-Pressure Response of Amyloid Folds.Viruses. 2019 Feb 28;11(3):202. doi: 10.3390/v11030202. Viruses. 2019. PMID: 30823361 Free PMC article. Review.
-
"What Doesn't Kill You Makes You Stronger": Future Applications of Amyloid Aggregates in Biomedicine.Molecules. 2020 Nov 11;25(22):5245. doi: 10.3390/molecules25225245. Molecules. 2020. PMID: 33187056 Free PMC article. Review.
-
Early events in light chain aggregation at physiological pH reveal new insights on assembly, stability, and aggregate dissociation.Amyloid. 2021 Jun;28(2):113-124. doi: 10.1080/13506129.2021.1877129. Epub 2021 Feb 3. Amyloid. 2021. PMID: 33533277 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

