CSF-induced and HIV-1-mediated distinct regulation of Hck and C/EBPbeta represent a heterogeneous susceptibility of monocyte-derived macrophages to M-tropic HIV-1 infection
- PMID: 12900520
- PMCID: PMC2194092
- DOI: 10.1084/jem.20022018
CSF-induced and HIV-1-mediated distinct regulation of Hck and C/EBPbeta represent a heterogeneous susceptibility of monocyte-derived macrophages to M-tropic HIV-1 infection
Abstract
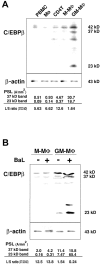
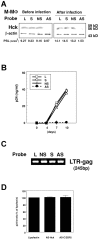
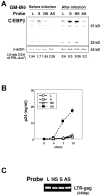
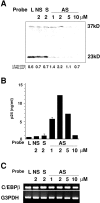
Granulocyte/macrophage colony-stimulating factor (GM-CSF)-induced monocyte-derived macrophages (GM-MPhi) are permissive to M-tropic HIV-1 entry, but inhibit viral replication at posttranscriptional and translational levels, whereas M-CSF-induced macrophages (M-MPhi) produce a large amount of HIV-1. M-MPhi express a high level of Hck and a large isoform of C/EBPbeta, and HIV-1 infection increases the expression of Hck but not of C/EBPbeta. GM-MPhi express a high level of C/EBPbeta and a low level of Hck, and HIV-1 infection drastically increases the expression of a short isoform of C/EBPbeta but decreases that of Hck. Treatment of M-MPhi with antisense oligonucleotide for Hck (AS-Hck) not only suppresses the expression of Hck, but also stimulates the induction of the short isoform of C/EBPbeta and inhibits the viral replication. Treatment of GM-MPhi with a moderate amount of AS-C/EBPbeta not only inhibits the expression of the small isoform of C/EBPbeta preferentially, but also stimulates the induction of Hck and stimulates the virus production at a high rate. These results suggest that CSF-induced and HIV-1-mediated distinct regulation of Hck and small isoform of C/EBPbeta represent the heterogeneous susceptibility of tissue MPhi to HIV-1 infection, and the regulation of Hck and C/EBPbeta are closely related and these two molecules affect one another.
Figures



Similar articles
-
Erythromycin derivatives inhibit HIV-1 replication in macrophages through modulation of MAPK activity to induce small isoforms of C/EBPbeta.Proc Natl Acad Sci U S A. 2008 Aug 26;105(34):12509-14. doi: 10.1073/pnas.0805504105. Epub 2008 Aug 21. Proc Natl Acad Sci U S A. 2008. PMID: 18719105 Free PMC article.
-
Suppression of HIV replication in human monocyte-derived macrophages induced by granulocyte/macrophage colony-stimulating factor.AIDS Res Hum Retroviruses. 1995 Sep;11(9):1031-8. doi: 10.1089/aid.1995.11.1031. AIDS Res Hum Retroviruses. 1995. PMID: 8554900
-
Catalase plays a critical role in the CSF-independent survival of human macrophages via regulation of the expression of BCL-2 family.J Biol Chem. 2005 Dec 16;280(50):41137-45. doi: 10.1074/jbc.M509793200. Epub 2005 Oct 4. J Biol Chem. 2005. PMID: 16204228
-
Effect of GM-CSF on HIV-1 replication in monocytes/macrophages in vivo and in vitro: a review.Vet Immunol Immunopathol. 1998 May 15;63(1-2):111-21. doi: 10.1016/s0165-2427(98)00087-7. Vet Immunol Immunopathol. 1998. PMID: 9656446 Review.
-
Functional heterogeneity of colony-stimulating factor-induced human monocyte-derived macrophages.Int J Hematol. 2002 Jul;76(1):27-34. doi: 10.1007/BF02982715. Int J Hematol. 2002. PMID: 12138892 Review.
Cited by
-
M-CSF increases proliferation and phagocytosis while modulating receptor and transcription factor expression in adult human microglia.J Neuroinflammation. 2013 Jul 17;10:85. doi: 10.1186/1742-2094-10-85. J Neuroinflammation. 2013. PMID: 23866312 Free PMC article.
-
Human immunodeficiency virus infection inhibits granulocyte-macrophage colony-stimulating factor-induced microglial proliferation.J Neurovirol. 2007 Dec;13(6):536-48. doi: 10.1080/13550280701549417. J Neurovirol. 2007. PMID: 18097885
-
Effect of apoptotic cell recognition on macrophage polarization and mycobacterial persistence.Infect Immun. 2014 Sep;82(9):3968-78. doi: 10.1128/IAI.02194-14. Epub 2014 Jul 14. Infect Immun. 2014. PMID: 25024361 Free PMC article.
-
Allosteric loss-of-function mutations in HIV-1 Nef from a long-term non-progressor.J Mol Biol. 2007 Nov 16;374(1):121-9. doi: 10.1016/j.jmb.2007.09.009. Epub 2007 Sep 11. J Mol Biol. 2007. PMID: 17920628 Free PMC article.
-
Macrophage colony-stimulating factor in the pathogenesis of HIV infection: potential target for therapeutic intervention.J Neuroimmune Pharmacol. 2006 Mar;1(1):32-40. doi: 10.1007/s11481-005-9003-1. J Neuroimmune Pharmacol. 2006. PMID: 18040789 Review.
References
-
- Lane, B.R., D.M. Markovitz, N.L. Woodford, R. Rochford, R.M. Strieter, and M.J. Coffey. 1999. TNF-alpha inhibits HIV-1 replication in peripheral blood monocytes and alveolar macrophages by inducing the production of RANTES and decreasing C-C chemokine receptor 5 (CCR5) expression. J. Immunol. 163:3653–3661. - PubMed
-
- Chun, T.W., L. Carruth, D. Finzi, X. Shen, J.A. DiGiuseppe, H. Taylor, M. Hermankova, K. Chadwick, J. Margolick, T.C. Quinn, et al. 1997. Quantification of latent tissue reservoirs and total body viral load in HIV-1 infection. Nature. 387:183–188. - PubMed
-
- Li, S., J. Juarez, M. Alali, D. Dwyer, R. Collman, A. Cunningham, and H.M. Naif. 1999. Persistent CCR5 utilization and enhanced macrophage tropism by primary blood human immunodeficiency virus type 1 isolates from advanced stages of disease and comparison to tissue-derived isolates. J. Virol. 73:9741–9755. - PMC - PubMed
-
- Hatch, W.C., A.R. Freedman, D.M. Boldt-Houle, J.E. Groopman, and E.F. Terwilliger. 1997. Differential effects of interleukin-13 on cytomegalovirus and human immunodeficiency virus infection in human alveolar macrophages. Blood. 89:3443–3450. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

