Vibrio harveyi nitroreductase is also a chromate reductase
- PMID: 12902220
- PMCID: PMC169119
- DOI: 10.1128/AEM.69.8.4390-4395.2003
Vibrio harveyi nitroreductase is also a chromate reductase
Abstract
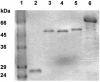
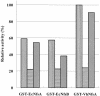
The chromate reductase purified from Pseudomonas ambigua was found to be homologous with several nitroreductases. Escherichia coli DH5alpha and Vibrio harveyi KCTC 2720 nitroreductases were chosen for the present study, and their chromate-reducing activities were determined. A fusion between glutathione S-transferase (GST) and E. coli DH5alpha NfsA (GST-EcNfsA), a fusion between GST and E. coli DH5alpha NfsB (GST-EcNfsB), and a fusion between GST and V. harveyi KCTC 2720 NfsA (GST-VhNfsA) were prepared for their overproduction and easy purification. GST-EcNfsA, GST-EcNFsB, and GST-VhNFsA efficiently reduced nitrofurazone and 2,4,6-trinitrotoluene (TNT) as their nitro substrates. The K(m) values for GST-EcNfsA, GST-EcNfsB, and GST-VhNfsA for chromate reduction were 11.8, 23.5, and 5.4 micro M, respectively. The V(max) values for GST-EcNfsA, GST-EcNfsB, and GST-VhNfsA were 3.8, 3.9, and 10.7 nmol/min/mg of protein, respectively. GST-VhNfsA was the most effective of the three chromate reductases, as determined by each V(max)/K(m) value. The optimal temperatures of GST-EcNfsA, GST-EcNfsB, and GST-VhNfsA for chromate reduction were 55, 30, and 30 degrees C, respectively. Thus, it is confirmed that nitroreductase can also act as a chromate reductase. Nitroreductases may be used in chromate remediation. GST-EcNfsA, GST-EcNfsB, and GST-VhNfsA have a molecular mass of 50 kDa and exist as a monomer in solution. Thin-layer chromatography showed that GST-EcNfsA, GST-EcNfsB, and GST-VhNfsA contain FMN as a cofactor. GST-VhNfsA reduced Cr(VI) to Cr(III). Cr(III) was much less toxic to E. coli than Cr(VI).
Figures



Similar articles
-
Biochemical characterization of NfsA, the Escherichia coli major nitroreductase exhibiting a high amino acid sequence homology to Frp, a Vibrio harveyi flavin oxidoreductase.J Bacteriol. 1996 Aug;178(15):4508-14. doi: 10.1128/jb.178.15.4508-4514.1996. J Bacteriol. 1996. PMID: 8755878 Free PMC article.
-
Mechanism of chromate reduction by the Escherichia coli protein, NfsA, and the role of different chromate reductases in minimizing oxidative stress during chromate reduction.Environ Microbiol. 2004 Aug;6(8):851-60. doi: 10.1111/j.1462-2920.2004.00639.x. Environ Microbiol. 2004. PMID: 15250887
-
Conversion of NfsA, the major Escherichia coli nitroreductase, to a flavin reductase with an activity similar to that of Frp, a flavin reductase in Vibrio harveyi, by a single amino acid substitution.J Bacteriol. 1998 Jan;180(2):422-5. doi: 10.1128/JB.180.2.422-425.1998. J Bacteriol. 1998. PMID: 9440535 Free PMC article.
-
Discovery and characterization of the flavonoids in Cortex Mori Radicis as naturally occurring inhibitors against intestinal nitroreductases.Chem Biol Interact. 2022 Dec 1;368:110222. doi: 10.1016/j.cbi.2022.110222. Epub 2022 Oct 14. Chem Biol Interact. 2022. PMID: 36244406
-
Activity coupling and complex formation between bacterial luciferase and flavin reductases.Photochem Photobiol Sci. 2008 Feb;7(2):183-8. doi: 10.1039/b713462b. Epub 2007 Dec 20. Photochem Photobiol Sci. 2008. PMID: 18264585 Review.
Cited by
-
Escherichia coli NemA is an efficient chromate reductase that can be biologically immobilized to provide a cell free system for remediation of hexavalent chromium.PLoS One. 2013;8(3):e59200. doi: 10.1371/journal.pone.0059200. Epub 2013 Mar 13. PLoS One. 2013. PMID: 23527133 Free PMC article.
-
Metal-tolerant thermophiles: metals as electron donors and acceptors, toxicity, tolerance and industrial applications.Environ Sci Pollut Res Int. 2018 Feb;25(5):4105-4133. doi: 10.1007/s11356-017-0869-2. Epub 2017 Dec 14. Environ Sci Pollut Res Int. 2018. PMID: 29238927 Review.
-
Removal efficiency of Cr6+ by indigenous Pichia sp. isolated from textile factory effluent.ScientificWorldJournal. 2012;2012:708213. doi: 10.1100/2012/708213. Epub 2012 May 2. ScientificWorldJournal. 2012. PMID: 22629188 Free PMC article.
-
Genome-resolved metagenomics provides insights into the ecological roles of the keystone taxa in heavy-metal-contaminated soils.Front Microbiol. 2023 Jul 21;14:1203164. doi: 10.3389/fmicb.2023.1203164. eCollection 2023. Front Microbiol. 2023. PMID: 37547692 Free PMC article.
-
Tibet plateau probiotic mitigates chromate toxicity in mice by alleviating oxidative stress in gut microbiota.Commun Biol. 2020 May 15;3(1):242. doi: 10.1038/s42003-020-0968-3. Commun Biol. 2020. PMID: 32415160 Free PMC article.
References
-
- Banks, R. B., and R. T. Cooke, Jr. 1986. Chromate reduction by rabbit liver aldehyde oxidase. Biochem. Biophys. Res. Commun. 137:8-14. - PubMed
-
- Bhide, J. V., P. K. Dhakephallker, and K. M. Paknikar. 1996. Microbiological process for the removal of Cr(VI) from chromate-bearing cooling tower effluent. Biotechnol. Lett. 18:667-672.
-
- Bryant, D. W., D. R. McCalla, M. Leeksma, and P. Laneuville. 1981. Type I nitroreductases of Escherichia coli. Can. J. Microbiol. 27:81-86. - PubMed
-
- Campos, J., M. Martinez-Pacheco, and C. Cervantes. 1995. Hexavalent-chromium reduction by a chromate-resistant Bacillus sp. strain. Antonie Leeuwenhoek 68:203-208. - PubMed
-
- Cervantes, C., and S. Silver. 1992. Plasmid chromate resistance and chromate reduction. Plasmid 27:65-71. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

