The mode of action of the Bacillus thuringiensis vegetative insecticidal protein Vip3A differs from that of Cry1Ab delta-endotoxin
- PMID: 12902253
- PMCID: PMC169065
- DOI: 10.1128/AEM.69.8.4648-4657.2003
The mode of action of the Bacillus thuringiensis vegetative insecticidal protein Vip3A differs from that of Cry1Ab delta-endotoxin
Abstract
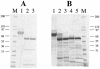
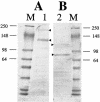
The Vip3A protein, secreted by Bacillus spp. during the vegetative stage of growth, represents a new family of insecticidal proteins. In our investigation of the mode of action of Vip3A, the 88-kDa Vip3A full-length toxin (Vip3A-F) was proteolytically activated to an approximately 62-kDa core toxin either by trypsin (Vip3A-T) or lepidopteran gut juice extracts (Vip3A-G). Biotinylated Vip3A-G demonstrated competitive binding to lepidopteran midgut brush border membrane vesicles (BBMV). Furthermore, in ligand blotting experiments with BBMV from the tobacco hornworm, Manduca sexta (Linnaeus), activated Cry1Ab bound to 120-kDa aminopeptidase N (APN)-like and 250-kDa cadherin-like molecules, whereas Vip3A-G bound to 80-kDa and 100-kDa molecules which are distinct from the known Cry1Ab receptors. In addition, separate blotting experiments with Vip3A-G did not show binding to isolated Cry1A receptors, such as M. sexta APN protein, or a cadherin Cry1Ab ecto-binding domain. In voltage clamping assays with dissected midgut from the susceptible insect, M. sexta, Vip3A-G clearly formed pores, whereas Vip3A-F was incapable of pore formation. In the same assay, Vip3A-G was incapable of forming pores with larvae of the nonsusceptible insect, monarch butterfly, Danaus plexippus (Linnaeus). In planar lipid bilayers, both Vip3A-G and Vip3A-T formed stable ion channels in the absence of any receptors, supporting pore formation as an inherent property of Vip3A. Both Cry1Ab and Vip3A channels were voltage independent and highly cation selective; however, they differed considerably in their principal conductance state and cation specificity. The mode of action of Vip3A supports its use as a novel insecticidal agent.
Figures



Similar articles
-
Specific epitopes of domains II and III of Bacillus thuringiensis Cry1Ab toxin involved in the sequential interaction with cadherin and aminopeptidase-N receptors in Manduca sexta.J Biol Chem. 2006 Nov 10;281(45):34032-9. doi: 10.1074/jbc.M604721200. Epub 2006 Sep 12. J Biol Chem. 2006. PMID: 16968705
-
Single amino acid changes in domain II of Bacillus thuringiensis CryIAb delta-endotoxin affect irreversible binding to Manduca sexta midgut membrane vesicles.J Bacteriol. 1995 May;177(9):2276-82. doi: 10.1128/jb.177.9.2276-2282.1995. J Bacteriol. 1995. PMID: 7730254 Free PMC article.
-
Role of alkaline phosphatase from Manduca sexta in the mechanism of action of Bacillus thuringiensis Cry1Ab toxin.J Biol Chem. 2010 Apr 23;285(17):12497-503. doi: 10.1074/jbc.M109.085266. Epub 2010 Feb 22. J Biol Chem. 2010. PMID: 20177063 Free PMC article.
-
Bacillus thuringiensis vegetative insecticidal protein family Vip3A and mode of action against pest Lepidoptera.Pest Manag Sci. 2020 May;76(5):1612-1617. doi: 10.1002/ps.5804. Epub 2020 Mar 11. Pest Manag Sci. 2020. PMID: 32103608 Review.
-
Current models of the mode of action of Bacillus thuringiensis insecticidal crystal proteins: a critical review.J Invertebr Pathol. 2012 Sep 15;111(1):1-12. doi: 10.1016/j.jip.2012.05.001. Epub 2012 May 19. J Invertebr Pathol. 2012. PMID: 22617276 Review.
Cited by
-
Analysis of Cry1Ah Toxin-Binding Reliability to Midgut Membrane Proteins of the Asian Corn Borer.Toxins (Basel). 2020 Jun 24;12(6):418. doi: 10.3390/toxins12060418. Toxins (Basel). 2020. PMID: 32599715 Free PMC article.
-
Transgenic soybean expressing Cry1Ab-Vip3A fusion protein confers broad-spectrum resistance to lepidopteran pest.Plant Cell Rep. 2025 Mar 25;44(4):87. doi: 10.1007/s00299-025-03463-4. Plant Cell Rep. 2025. PMID: 40131492 Free PMC article.
-
Molecular architecture and activation of the insecticidal protein Vip3Aa from Bacillus thuringiensis.Nat Commun. 2020 Aug 7;11(1):3974. doi: 10.1038/s41467-020-17758-5. Nat Commun. 2020. PMID: 32769995 Free PMC article.
-
Activity of Vip3Aa1 against Periplaneta Americana.Open Life Sci. 2020 Mar 24;15:133-144. doi: 10.1515/biol-2020-0014. eCollection 2020. Open Life Sci. 2020. PMID: 33987470 Free PMC article.
-
Impact of Temperature Change on the Fall Armyworm, Spodoptera frugiperda under Global Climate Change.Insects. 2022 Oct 26;13(11):981. doi: 10.3390/insects13110981. Insects. 2022. PMID: 36354805 Free PMC article. Review.
References
-
- Aronson, A. I., and Y. Shai. 2001. Why Bacillus thuringiensis toxins are so effective: unique features of their mode of action. FEBS Microbiol. Lett. 195:1-8. - PubMed
-
- Dean, D. H., F. Rajamohan, M. K. Lee, S.-J. Wu, X. J. Chen, and E. Alcantara. 1996. Probing the mechanism of action of Bacillus thuringiensis insecticidal proteins by site-directed mutagenesis—a minireview. Gene 179:111-117. - PubMed
-
- Denolf, P., K. Hendrickx, J. Van Damme, S. Jansens, M. Peferoen, D. Degheele, and J. Van Rie. 1997. Cloning and characterization of Manduca sexta and Plutella xylostella midgut aminopeptidase N enzymes related to Bacillus thuringiensis toxin-binding proteins. Eur. J. Biochem. 248:748-761. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

