Alteration of chain length substrate specificity of Aeromonas caviae R-enantiomer-specific enoyl-coenzyme A hydratase through site-directed mutagenesis
- PMID: 12902277
- PMCID: PMC169158
- DOI: 10.1128/AEM.69.8.4830-4836.2003
Alteration of chain length substrate specificity of Aeromonas caviae R-enantiomer-specific enoyl-coenzyme A hydratase through site-directed mutagenesis
Abstract
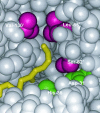
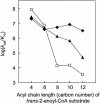
Aeromonas caviae R-specific enoyl-coenzyme A (enoyl-CoA) hydratase (PhaJ(Ac)) is capable of providing (R)-3-hydroxyacyl-CoA with a chain length of four to six carbon atoms from the fatty acid beta-oxidation pathway for polyhydroxyalkanoate (PHA) synthesis. In this study, amino acid substitutions were introduced into PhaJ(Ac) by site-directed mutagenesis to investigate the feasibility of altering the specificity for the acyl chain length of the substrate. A crystallographic structure analysis of PhaJ(Ac) revealed that Ser-62, Leu-65, and Val-130 define the width and depth of the acyl-chain-binding pocket. Accordingly, we targeted these three residues for amino acid substitution. Nine single-mutation enzymes and two double-mutation enzymes were generated, and their hydratase activities were assayed in vitro by using trans-2-octenoyl-CoA (C(8)) as a substrate. Three of these mutant enzymes, L65A, L65G, and V130G, exhibited significantly high activities toward octenoyl-CoA than the wild-type enzyme exhibited. PHA formation from dodecanoate (C(12)) was examined by using the mutated PhaJ(Ac) as a monomer supplier in recombinant Escherichia coli LS5218 harboring a PHA synthase gene from Pseudomonas sp. strain 61-3 (phaC1(Ps)). When L65A, L65G, or V130G was used individually, increased molar fractions of 3-hydroxyoctanoate (C(8)) and 3-hydroxydecanoate (C(10)) units were incorporated into PHA. These results revealed that Leu-65 and Val-130 affect the acyl chain length substrate specificity. Furthermore, comparative kinetic analyses of the wild-type enzyme and the L65A and V130G mutants were performed, and the mechanisms underlying changes in substrate specificity are discussed.
Figures

Similar articles
-
Site-directed mutagenesis of Aeromonas hydrophila enoyl coenzyme A hydratase enhancing 3-hydroxyhexanoate fractions of poly(3-hydroxybutyrate-co-3-hydroxyhexanoate).Curr Microbiol. 2007 Jul;55(1):20-4. doi: 10.1007/s00284-006-0490-y. Epub 2007 May 28. Curr Microbiol. 2007. PMID: 17534562
-
Expression and characterization of (R)-specific enoyl coenzyme A hydratase involved in polyhydroxyalkanoate biosynthesis by Aeromonas caviae.J Bacteriol. 1998 Feb;180(3):667-73. doi: 10.1128/JB.180.3.667-673.1998. J Bacteriol. 1998. PMID: 9457873 Free PMC article.
-
Molecular cloning of two (R)-specific enoyl-CoA hydratase genes from Pseudomonas aeruginosa and their use for polyhydroxyalkanoate synthesis.FEMS Microbiol Lett. 2000 Mar 15;184(2):193-8. doi: 10.1111/j.1574-6968.2000.tb09013.x. FEMS Microbiol Lett. 2000. PMID: 10713420
-
Crystal structure of the (R)-specific enoyl-CoA hydratase from Aeromonas caviae involved in polyhydroxyalkanoate biosynthesis.J Biol Chem. 2003 Jan 3;278(1):617-24. doi: 10.1074/jbc.M205484200. Epub 2002 Oct 29. J Biol Chem. 2003. PMID: 12409309
-
Enoyl-CoA hydratase. reaction, mechanism, and inhibition.Bioorg Med Chem. 2003 Jan 2;11(1):9-20. doi: 10.1016/s0968-0896(02)00333-4. Bioorg Med Chem. 2003. PMID: 12467702 Review.
Cited by
-
Protein design for pathway engineering.J Struct Biol. 2014 Feb;185(2):234-42. doi: 10.1016/j.jsb.2013.03.011. Epub 2013 Apr 1. J Struct Biol. 2014. PMID: 23558037 Free PMC article. Review.
-
Site-directed mutagenesis of Aeromonas hydrophila enoyl coenzyme A hydratase enhancing 3-hydroxyhexanoate fractions of poly(3-hydroxybutyrate-co-3-hydroxyhexanoate).Curr Microbiol. 2007 Jul;55(1):20-4. doi: 10.1007/s00284-006-0490-y. Epub 2007 May 28. Curr Microbiol. 2007. PMID: 17534562
-
Structure and function of Rv0130, a conserved hypothetical protein from Mycobacterium tuberculosis.Protein Sci. 2006 Oct;15(10):2300-9. doi: 10.1110/ps.062309306. Epub 2006 Sep 8. Protein Sci. 2006. PMID: 16963641 Free PMC article.
-
Cocrystallization and preliminary crystallographic analysis of an inactive MaoC-like hydratase mutant with the substrate crotonyl-CoA.Acta Crystallogr Sect F Struct Biol Cryst Commun. 2012 Apr 1;68(Pt 4):483-5. doi: 10.1107/S1744309112007671. Epub 2012 Mar 28. Acta Crystallogr Sect F Struct Biol Cryst Commun. 2012. PMID: 22505426 Free PMC article.
-
Structure reveals regulatory mechanisms of a MaoC-like hydratase from Phytophthora capsici involved in biosynthesis of polyhydroxyalkanoates (PHAs).PLoS One. 2013 Nov 11;8(11):e80024. doi: 10.1371/journal.pone.0080024. eCollection 2013. PLoS One. 2013. PMID: 24244597 Free PMC article.
References
-
- Abe, H., Y. Doi, T. Fukushima, and H. Eya. 1994. Biosynthesis from gluconate of a random copolyester consisting of 3-hydroxybutyrate and medium-chain-length 3-hydroxyalkanoates by Pseudomonas sp. 61-3. Int. J. Biol. Macromol. 16:115-119. - PubMed
-
- Amara, A. A., A. Steinbüchel, and B. H. Rehm. 2002. In vivo evolution of the Aeromonas punctata polyhydroxyalkanoate (PHA) synthase: isolation and characterization of modified PHA synthases with enhanced activity. Appl. Microbiol. Biotechnol. 59:477-482. - PubMed
-
- Fiedler, S., A. Steinbüchel, and B. H. Rehm. 2002. The role of the fatty acid β-oxidation multienzyme complex from Pseudomonas oleovorans in polyhydroxyalkanoate biosynthesis: molecular characterization of the fadBA operon from P. oleovorans of the enoyl-CoA hydratase genes phaJ from P. oleovorans and Pseudomonas putida. Arch. Microbiol. 178:149-160. - PubMed
-
- Fong, J. C., and H. Schulz. 1981. Short-chain and long-chain enoyl-CoA hydratase from pig heart muscle. Methods Enzymol. 70:390-398. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

