Reversible paired helical filament-like phosphorylation of tau is an adaptive process associated with neuronal plasticity in hibernating animals
- PMID: 12904458
- PMCID: PMC6740664
- DOI: 10.1523/JNEUROSCI.23-18-06972.2003
Reversible paired helical filament-like phosphorylation of tau is an adaptive process associated with neuronal plasticity in hibernating animals
Abstract
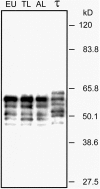
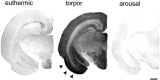
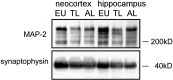
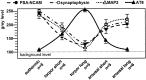
Neurofibrillary pathology [paired helical filaments (PHFs)] formed by the microtubule-associated protein tau in a hyperphosphorylated form is a major hallmark of Alzheimer's disease and related disorders. The process of tau phosphorylation, thought to be of critical importance for PHF formation, and its potential link to neurodegeneration, however, is not understood very well, mostly because of the lack of a physiological in vivo model of PHF-like tau phosphorylation. Here we describe the formation of highly phosphorylated tau, containing a number of PHF-like epitopes in torpor during hibernation. PHF-like phosphorylation of tau was not associated with fibril formation and was fully reversible after arousal. Distribution of PHF-like tau followed a consistent pattern, being most intense in the entorhinal cortex, hippocampus, and isocortical areas. Within the hippocampus, a particularly high labeling was seen in CA3 pyramidal cells. Somewhat lesser reactivity was present in CA1 neurons while dentate gyrus granule cells were not reactive. Formation of PHF-like tau in CA3 neurons was paralleled by the regression of synaptic contacts of the mossy fiber system terminating on CA3 apical dendrites. Mossy fiber afferentation was re-established during arousal, concomitantly with the decrease of PHF-like tau in CA3 neurons. These findings implicate an essential link between neuronal plasticity and PHF-like phosphorylation of tau. The repeated formation and degradation of PHF-like tau might, thus, represent a physiological mechanism not necessarily associated with pathological effects. Hibernation will, therefore, be a valuable model to study the regulation of PHF-like tau-phosphorylation and its cell biological sequelae under physiological in vivo conditions.
Figures




References
-
- Alexander L ( 1934) The neurofibrils in systemic disease and in supravital experiments, with remarks on pseudo-atrophy of the brain. Arch Neurol Psychiatry 32: 933-962.
-
- Arendt T, Brückner MK, Gertz HJ, Marcova L ( 1998a) Cortical distribution of neurofibrillary tangles in Alzheimer's disease matches the pattern of neurones that retain their capacity of plastic remodelling in the adult brain. Neuroscience 83: 991-1002. - PubMed
-
- Arendt T, Holzer M, Fruth R, Brückner MK, Gärtner U ( 1998b) Phosphorylation of tau, Aβ-formation, and apoptosis after in vivo inhibition of PP-1 and PP-2A. Neurobiol Aging 19: 3-13. - PubMed
-
- Armstrong SC, Ganote CE ( 1992) Effects of protein phosphatase inhibitors okadaic acid and calyculin A on metabolically inhibited and ischaemic isolated myocytes. J Mol Cell Cardiol 24: 869-884. - PubMed
-
- Ashford JW, Jarvik L ( 1985) Alzheimer's disease: does neuron plasticity pre-dispose to axonal neurofibrillary degeneration? N Engl J Med 313: 388-389. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
