Nod factor-induced root hair curling: continuous polar growth towards the point of nod factor application
- PMID: 12913154
- PMCID: PMC181283
- DOI: 10.1104/pp.103.021634
Nod factor-induced root hair curling: continuous polar growth towards the point of nod factor application
Abstract
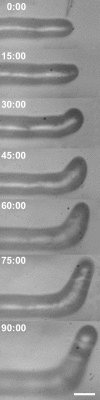
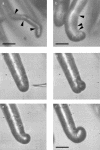
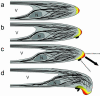
A critical step in establishing a successful nitrogen-fixing symbiosis between rhizobia and legume plants is the entrapment of the bacteria between root hair cell walls, usually in characteristic 180 degrees to 360 degrees curls, shepherd's crooks, which are formed by the host's root hairs. Purified bacterial signal molecules, the nodulation factors (NFs), which are lipochitooligosaccharides, induce root hair deformation in the appropriate host legume and have been proposed to be a key player in eliciting root hair curling. However, for curling to occur, the presence of intact bacteria is thought to be essential. Here, we show that, when spot applied to one side of the growing Medicago truncatula root hair tip, purified NF alone is sufficient to induce reorientation of the root hair growth direction, or a full curl. Using wild-type M. truncatula containing the pMtENOD11::GUS construct, we demonstrate that MtENOD11::GUS is expressed after spot application. The data have been incorporated into a cell biological model, which explains the formation of shepherd's crook curls around NF-secreting rhizobia by continuous tip growth reorientation.
Figures


Similar articles
-
The small GTPase ROP10 of Medicago truncatula is required for both tip growth of root hairs and nod factor-induced root hair deformation.Plant Cell. 2015 Mar;27(3):806-22. doi: 10.1105/tpc.114.135210. Epub 2015 Mar 20. Plant Cell. 2015. PMID: 25794934 Free PMC article.
-
A nonsymbiotic root hair tip growth phenotype in NORK-mutated legumes: implications for nodulation factor-induced signaling and formation of a multifaceted root hair pocket for bacteria.Plant Cell. 2004 Apr;16(4):933-44. doi: 10.1105/tpc.019653. Epub 2004 Mar 18. Plant Cell. 2004. PMID: 15031407 Free PMC article.
-
The NFP locus of Medicago truncatula controls an early step of Nod factor signal transduction upstream of a rapid calcium flux and root hair deformation.Plant J. 2003 May;34(4):495-506. doi: 10.1046/j.1365-313x.2003.01743.x. Plant J. 2003. PMID: 12753588
-
Biological activity of Nod factors.Acta Biochim Pol. 2020 Oct 22;67(4):435-440. doi: 10.18388/abp.2020_5353. Acta Biochim Pol. 2020. PMID: 33090749 Review.
-
Leguminous plants: inventors of root nodules to accommodate symbiotic bacteria.Int Rev Cell Mol Biol. 2015;316:111-58. doi: 10.1016/bs.ircmb.2015.01.004. Epub 2015 Feb 20. Int Rev Cell Mol Biol. 2015. PMID: 25805123 Review.
Cited by
-
Mass Spectrometric-Based Selected Reaction Monitoring of Protein Phosphorylation during Symbiotic Signaling in the Model Legume, Medicago truncatula.PLoS One. 2016 May 20;11(5):e0155460. doi: 10.1371/journal.pone.0155460. eCollection 2016. PLoS One. 2016. PMID: 27203723 Free PMC article.
-
Phosphate-deprivation and damage signalling by extracellular ATP.Front Plant Sci. 2023 Jan 12;13:1098146. doi: 10.3389/fpls.2022.1098146. eCollection 2022. Front Plant Sci. 2023. PMID: 36714742 Free PMC article.
-
Microtubule array formation during root hair infection thread initiation and elongation in the Mesorhizobium-Lotus symbiosis.Protoplasma. 2014 Sep;251(5):1099-111. doi: 10.1007/s00709-014-0618-z. Epub 2014 Feb 2. Protoplasma. 2014. PMID: 24488109
-
Rapid phosphoproteomic and transcriptomic changes in the rhizobia-legume symbiosis.Mol Cell Proteomics. 2012 Sep;11(9):724-44. doi: 10.1074/mcp.M112.019208. Epub 2012 Jun 8. Mol Cell Proteomics. 2012. PMID: 22683509 Free PMC article.
-
A nuclear-targeted cameleon demonstrates intranuclear Ca2+ spiking in Medicago truncatula root hairs in response to rhizobial nodulation factors.Plant Physiol. 2009 Nov;151(3):1197-206. doi: 10.1104/pp.109.142851. Epub 2009 Aug 21. Plant Physiol. 2009. PMID: 19700563 Free PMC article.
References
-
- Ardourel M, Demont N, Debellé F, Maillet F, De Billy F, Promé J-C, Dénarié J, Truchet G (1994) Rhizobium meliloti lipooligosaccharide nodulation factors: different structural requirements for bacterial entry into target root hair cells and induction of plant symbiotic developmental responses. Plant Cell 6: 1357–1374 - PMC - PubMed
-
- Battey NH, Blackbourn HD (1993) The control of exocytosis in plant cells. New Phytol 125: 307–338 - PubMed
-
- Battey N, Carroll A, van Kesteren P, Taylor A, Brownlee C (1996) The measurement of exocytosis in plant cells. J Exp Bot 47: 717–728
-
- Cárdenas L, Feijó JA, Kunkel JG, Sánchez F, Holdaway-Clarke T, Hepler PK, Quinto C (1999) Rhizobium Nod factors induce increases in intracellular free calcium influxes in bean root hairs. Plant J 19: 347–352 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

