p16INK4A alterations are accompanied by aberrant protein immunostaining in endometrial carcinomas
- PMID: 12920579
- PMCID: PMC12161915
- DOI: 10.1007/s00432-003-0482-2
p16INK4A alterations are accompanied by aberrant protein immunostaining in endometrial carcinomas
Abstract
Purpose: To date, the significance of p16INK4A tumor suppressor gene inactivation in sporadic endometrial cancer (EC) has only rarely been described. In this study, we examined the alteration type and frequency of gene alterations [point mutations, aberrant promoter methylation and loss of heterozygosity (LOH)] in 50 sporadic ECs, and correlated the genetic findings with the immunohistochemical expression of the p16INK4A protein and the classical clinicopathological features.
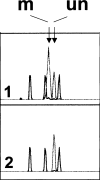
Methods: Gene mutations were detected by PCR-SSCP-sequencing analysis, promoter hypermethylation by methylation-specific PCR (MSP), and LOH by PCR of the STS-marker c5.1.
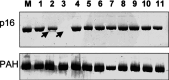
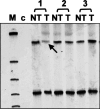
Results: In total, p16INK4A alterations were found in 14 of 50 (28%) sporadic ECs. In six (12%) cases, two alterations occurred simultaneously. Partial p16INK4A deletions were found in four of 50 (8%) samples. There was one missense mutation (codon 70; CCC-->GCC) and one frameshift mutation (1-bp deletion in exon 2). Only 2 of 47 (4.2%) tumors exhibited aberrant promoter methylation. An allelic loss was detected in 12 of 50 (24%) carcinomas with a higher incidence in advanced endometrial carcinomas than in early-stage uterine tumors. p16INK4A alterations were generally accompanied by gene silencing, confirmed by aberrant protein immunostaining ( r=-0.442; P=0.001). There was a significant difference in the frequency of p16INK4A alterations between early (stage I; 18%) and advanced (stages II-IV; 58%) ECs ( P=0.022). One case showed complete protein loss, but absence of genetic alterations.
Conclusions: Our data indicate that p16INK4A inactivation plays a role in the tumorigenesis of the subset of sporadic ECs, particularly in cases exhibiting an aggressive clinical behavior. We demonstrate that p16INK4A methylation can act efficiently and similarly to other genetic alterations as one of the two necessary hits according to the Knudson two-hit hypothesis of tumor suppressor gene inactivation.
Figures
Similar articles
-
Bcl-2 is closely correlated with favorable prognostic factors and inversely associated with p53 protein accumulation in endometrial carcinomas: immunohistochemical and polymerase chain reaction/loss of heterozygosity findings.J Cancer Res Clin Oncol. 1997;123(8):429-34. doi: 10.1007/BF01372546. J Cancer Res Clin Oncol. 1997. PMID: 9292705 Free PMC article.
-
Aberrant promoter methylation and inactivation of PTEN gene in cervical carcinoma from Indian population.J Cancer Res Clin Oncol. 2011 Aug;137(8):1255-62. doi: 10.1007/s00432-011-0994-0. Epub 2011 Jun 23. J Cancer Res Clin Oncol. 2011. PMID: 21698421 Free PMC article.
-
Diagnostic test accuracy and cost-effectiveness of tests for codeletion of chromosomal arms 1p and 19q in people with glioma.Cochrane Database Syst Rev. 2022 Mar 2;3(3):CD013387. doi: 10.1002/14651858.CD013387.pub2. Cochrane Database Syst Rev. 2022. PMID: 35233774 Free PMC article.
-
The pattern of p14ARF expression in primary and metastatic human endometrial carcinomas: correlation with clinicopathological features and TP53 pathway alterations.Int J Gynecol Cancer. 2010 Aug;20(6):993-9. doi: 10.1111/IGC.0b013e3181e76a4d. Int J Gynecol Cancer. 2010. PMID: 20683407
-
Electronic cigarettes for smoking cessation.Cochrane Database Syst Rev. 2022 Nov 17;11(11):CD010216. doi: 10.1002/14651858.CD010216.pub7. Cochrane Database Syst Rev. 2022. Update in: Cochrane Database Syst Rev. 2024 Jan 8;1:CD010216. doi: 10.1002/14651858.CD010216.pub8. PMID: 36384212 Free PMC article. Updated.
Cited by
-
Recent findings on epigenetic gene abnormalities involved in uterine cancer.Mol Clin Oncol. 2017 Nov;7(5):733-737. doi: 10.3892/mco.2017.1428. Epub 2017 Sep 20. Mol Clin Oncol. 2017. PMID: 29181164 Free PMC article.
-
PRMT5 promotes progression of endometrioid adenocarcinoma via ERα and cell cycle signaling pathways.J Pathol Clin Res. 2021 Mar;7(2):154-164. doi: 10.1002/cjp2.194. Epub 2021 Jan 8. J Pathol Clin Res. 2021. PMID: 33416213 Free PMC article.
-
DNA methylation in endometrial cancer.Epigenetics. 2010 Aug 16;5(6):491-8. doi: 10.4161/epi.5.6.12431. Epub 2010 Aug 16. Epigenetics. 2010. PMID: 20543579 Free PMC article. Review.
-
The genomics and genetics of endometrial cancer.Adv Genomics Genet. 2012 Mar;2012(2):33-47. doi: 10.2147/AGG.S28953. Adv Genomics Genet. 2012. PMID: 22888282 Free PMC article.
-
hMLH1 promoter hypermethylation and MSI status in human endometrial carcinomas with and without metastases.Clin Exp Metastasis. 2012 Dec;29(8):889-900. doi: 10.1007/s10585-012-9478-0. Epub 2012 May 3. Clin Exp Metastasis. 2012. PMID: 22552371
References
-
- Caca K, Feisthammel J, Klee K, et al. (2002) Inactivation of the INK4a/ARF locus and p53 in sporadic extrahepatic bile duct cancers and bile tract cancer cell lines. Int J Cancer 97:481–488 - PubMed
-
- Cairns P, Polascik TJ, Eby Y, et al. (1995) Frequency of homozygous deletion at p16/CDKN2 in primary human tumours. Nat Genet 112:210–212 - PubMed
-
- Caldas C, Hahn SD, da Costa LT, et al. (1994) Frequent somatic mutations and homozygous deletions of the p16 (MTS1) gene in pancreatic adenocarcinoma. Nat Genet 8:27–32 - PubMed
-
- Coleman KG, Wautler BS, Morrisey D, et al. (1997) Identification of CDK4 sequences involved in cyclin D1 and p16 binding. J Biol Chem 272:18869–18874 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

