Halogenation of a capsaicin analogue leads to novel vanilloid TRPV1 receptor antagonists
- PMID: 12922928
- PMCID: PMC1573981
- DOI: 10.1038/sj.bjp.0705387
Halogenation of a capsaicin analogue leads to novel vanilloid TRPV1 receptor antagonists
Erratum in
- Br J Pharmacol. 2003 Nov;140(5):1008
Abstract
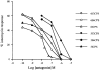
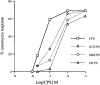
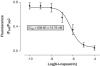
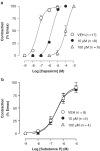
1. The C-5 halogenation of the vanillyl moiety of resiniferatoxin, an ultrapotent agonist of vanilloid TRPV1 receptors, results in a potent antagonist for these receptors. Here, we have synthesized a series of halogenated derivatives of 'synthetic capsaicin' (nonanoyl vanillamide=nordihydrocapsaicin) differing for the nature (iodine, bromine-chlorine) and the regiochemistry (C-5, C-6) of the halogenation. 2. The activity of these compounds was investigated on recombinant human TRPV1 receptors overexpressed in HEK-293 cells. None of the six compounds exerted any significant agonist activity, as assessed by measuring their effect on TRPV1-mediated calcium mobilization. Instead, all compounds antagonized, to various extents, the effect of capsaicin in this assay. 3. All 6-halo-nordihydrocapsaicins behaved as competitive antagonists against human TRPV1 according to the corresponding Schild's plots, and were more potent than the corresponding 5-halogenated analogues. The iodo-derivatives were more potent than the bromo- and chloro-derivatives. 4. Using human recombinant TRPV1, 6-iodo-nordihydrocapsaicin (IC(50)=10 nM against 100 nM capsaicin) was about four times more potent than the prototypical TRPV1 antagonist, capsazepine, and was tested against capsaicin also on native TRPV1 in: (i) rat dorsal root ganglion neurons in culture; (ii) guinea-pig urinary bladder; and (iii) guinea-pig bronchi. In all cases, except for the guinea-pig bronchi, the compound was significantly more potent than capsazepine as a TRPV1 antagonist. 5. In conclusion, 6-iodo-nordihydrocapsaicin, a stable and easily prepared compound, is a potent TRPV1 antagonist and a convenient replacement for capsazepine in most of the in vitro preparations currently used to assess the activity of putative vanilloid receptor agonists.
Figures


References
-
- BIRDER L.A., NAKAMURA Y., KISS S., NEALEN M.L., BARRICK S., KANAI A.J., WANG E., RUIZ G., DE GROAT W.C., APODACA G., WATKINS S., CATERINA M.J. Altered urinary bladder function in mice lacking the vanilloid receptor TRPV1. Nat. Neurosci. 2002;5:856–860. - PubMed
-
- CATERINA M.J., LEFFLER A., MALMBERG A.B., MARTIN W.J., TRAFTON J., PETERSEN-ZEITZ K.R., KOLTZENBURG M., BASBAUM A.I., JULIUS D. Impaired nociception and pain sensation in mice lacking the capsaicin receptor. Science. 2000;288:306–313. - PubMed
-
- CHUNG K.F., CHANG A.B. Therapy for cough: active agents. Pulm. Pharmacol. Ther. 2002;15:335–338. - PubMed
-
- DAVIS J.B., GRAY J., GUNTHORPE M.J., HATCHER J.P., DAVEY P.T., OVEREND P., HARRIES M.H., LATCHAM J., CLAPHAM C., ATKINSON K., HUGHES S.A., RANCE K., GRAU E., HARPER A.J., PUGH P.L., ROGERS D.C., BINGHAM S., RANDALL A., SHEARDOWN S.A. Vanilloid receptor-1 is essential for inflammatory thermal hyperalgesia. Nature. 2000;405:183–187. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

