Elasticity in extracellular matrix 'shape modules' of tendon, cartilage, etc. A sliding proteoglycan-filament model
- PMID: 12923209
- PMCID: PMC2343561
- DOI: 10.1113/jphysiol.2003.050179
Elasticity in extracellular matrix 'shape modules' of tendon, cartilage, etc. A sliding proteoglycan-filament model
Abstract
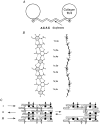
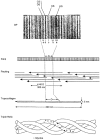
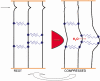
Connective tissues (CTs), which define bodily shape, must respond quickly, robustly and reversibly to deformations caused by internal and external stresses. Fibrillar (elastin, collagen) elasticity under tension depends on molecular and supramolecular mechanisms. A second intra-/inter-molecular pair, involving proteoglycans (PGs), is proposed to cope with compressive stresses. PG interfibrillar bridges ('shape modules'), supramolecular structures ubiquitously distributed throughout CT extracellular matrices (ECMs), are examined for potential elastic properties. L-iduronate residues in shape module decoran PGs are suggested to be molecular springs, cycling through alternative conformations. On a larger scale, anionic glycosaminoglycan (AGAG) interfibrillar bridges in shape modules are postulated to take part in a sliding filament (dashpot-like) process, which converts local compressions into disseminated tensile strains. The elasticity of fibrils and AGAGs, manifest at molecular and larger-scale levels, provides a graduated and smooth response to stresses of varying degrees. NMR and rheo NMR, computer modelling, electron histochemical, biophysical and chemical morphological evidence for the proposals is reviewed.
Figures

References
-
- Castellani PP, Moricutti M, Franchi M, Ruggeri A, Roveri N. Arrangement of microfibrils in collagen fibrils in the rat tail. Cell Tiss Res. 1983;234:735–743. - PubMed
-
- Casu B. Discussion. In: Scott JE, editor. Dermatan Sulphate Proteoglycans: Chemistry, Biology, Chemical Pathology. London: The Biochemical Society; 1993. p. p. 76.
-
- Casu B, Petitou M, Provasoli M, Sinay P. Conformational flexibility: a new concept for explaining binding and biological properties of iduronic acid-containing glycosaminoglycans. Trends Biochem Sci. 1988;13:221–225. - PubMed
-
- Davidson EA, Meyer K. Chondroitin, a new mucopolysaccharide. J Biol Chem. 1954;211:605–611. - PubMed
-
- Fatt I. Physiology of the eye. Boston and London: Butterworths; 1978. chap. 6.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

