Drosophila spalt/spalt-related mutants exhibit Townes-Brocks' syndrome phenotypes
- PMID: 12925729
- PMCID: PMC193554
- DOI: 10.1073/pnas.1836391100
Drosophila spalt/spalt-related mutants exhibit Townes-Brocks' syndrome phenotypes
Abstract
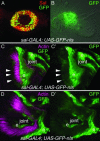
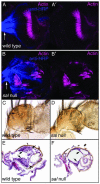
Mutations in SALL1, the human homolog of the Drosophila spalt gene, result in Townes-Brocks' syndrome, which is characterized by hand/foot, anogenital, renal, and ear anomalies, including sensorineural deafness. spalt genes encode zinc finger transcription factors that are found in animals as diverse as worms, insects, and vertebrates. Here, we examine the effect of losing both of the spalt genes, spalt and spalt-related, in the fruit fly Drosophila melanogaster, and report defects similar to those in humans with Townes-Brocks' syndrome. Loss of both spalt and spalt-related function in flies yields morphological defects in the testes, genitalia, and the antenna. Furthermore, spalt/spalt-related mutant antennae show severe reductions in Johnston's organ, the major auditory organ in Drosophila. Electrophysiological analyses confirm that spalt/spalt-related mutant flies are deaf. These commonalities suggest that there is functional conservation for spalt genes between vertebrates and insects.
Figures


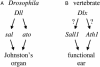
Similar articles
-
SALL1 mutations in Townes-Brocks syndrome and related disorders.Hum Mutat. 2000 Dec;16(6):460-6. doi: 10.1002/1098-1004(200012)16:6<460::AID-HUMU2>3.0.CO;2-4. Hum Mutat. 2000. PMID: 11102974 Review.
-
SALL1 mutation analysis in Townes-Brocks syndrome: twelve novel mutations and expansion of the phenotype.Hum Mutat. 2005 Sep;26(3):282. doi: 10.1002/humu.9362. Hum Mutat. 2005. PMID: 16088922
-
Distal-less and homothorax regulate multiple targets to pattern the Drosophila antenna.Development. 2002 Apr;129(8):1967-74. doi: 10.1242/dev.129.8.1967. Development. 2002. PMID: 11934862
-
High incidence of the R276X SALL1 mutation in sporadic but not familial Townes-Brocks syndrome and report of the first familial case.J Med Genet. 2003 Nov;40(11):e127. doi: 10.1136/jmg.40.11.e127. J Med Genet. 2003. PMID: 14627694 Free PMC article. No abstract available.
-
Regulation and function of Spalt proteins during animal development.Int J Dev Biol. 2009;53(8-10):1385-98. doi: 10.1387/ijdb.072408jd. Int J Dev Biol. 2009. PMID: 19247946 Review.
Cited by
-
The Drosophila auditory system.Wiley Interdiscip Rev Dev Biol. 2014 Mar-Apr;3(2):179-91. doi: 10.1002/wdev.128. Epub 2013 Oct 16. Wiley Interdiscip Rev Dev Biol. 2014. PMID: 24719289 Free PMC article. Review.
-
Sall genes regulate region-specific morphogenesis in the mouse limb by modulating Hox activities.Development. 2009 Feb;136(4):585-94. doi: 10.1242/dev.027748. Development. 2009. PMID: 19168674 Free PMC article.
-
Modeling congenital disease and inborn errors of development in Drosophila melanogaster.Dis Model Mech. 2016 Mar;9(3):253-69. doi: 10.1242/dmm.023564. Dis Model Mech. 2016. PMID: 26935104 Free PMC article. Review.
-
Cells, molecules and morphogenesis: the making of the vertebrate ear.Brain Res. 2006 May 26;1091(1):151-71. doi: 10.1016/j.brainres.2006.02.078. Epub 2006 Apr 27. Brain Res. 2006. PMID: 16643865 Free PMC article. Review.
-
The Spalt transcription factors regulate cell proliferation, survival and epithelial integrity downstream of the Decapentaplegic signalling pathway.Biol Open. 2013 Jan 15;2(1):37-48. doi: 10.1242/bio.20123038. Epub 2012 Oct 26. Biol Open. 2013. PMID: 23336075 Free PMC article.
References
-
- Kohlhase, J., Wischermann, A., Reichenbach, H., Froster, U. & Engel, W. (1998) Nat. Genet. 18, 81-83. - PubMed
-
- Kohlhase, J., Schuh, R., Dowe, G., Kuhnlein, R. P., Jackle, H., Schroeder, B., Schulz-Schaeffer, W., Kretzschmar, H. A., Kohler, A., Muller, U., et al. (1996) Genomics 38, 291-298. - PubMed
-
- Kohlhase, J., Hausmann, S., Stojmenovic, G., Dixkens, C., Bink, K., Schulz-Schaeffer, W., Altmann, M. & Engel, W. (1999) Genomics 62, 216-222. - PubMed
-
- Marlin, S., Blanchard, S., Slim, R., Lacombe, D., Denoyelle, F., Alessandri, J., Calzolari, E., Drouin-Garraud, V., Ferraz, F., Fourmaintraux, A., et al. (1999) Hum. Mutat. 14, 377-386. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

