Intense myocyte formation from cardiac stem cells in human cardiac hypertrophy
- PMID: 12928492
- PMCID: PMC193580
- DOI: 10.1073/pnas.1832855100
Intense myocyte formation from cardiac stem cells in human cardiac hypertrophy
Abstract
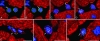
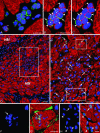
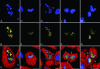
It is generally believed that increase in adult contractile cardiac mass can be accomplished only by hypertrophy of existing myocytes. Documentation of myocardial regeneration in acute stress has challenged this dogma and led to the proposition that myocyte renewal is fundamental to cardiac homeostasis. Here we report that in human aortic stenosis, increased cardiac mass results from a combination of myocyte hypertrophy and hyperplasia. Intense new myocyte formation results from the differentiation of stem-like cells committed to the myocyte lineage. These cells express stem cell markers and telomerase. Their number increased >13-fold in aortic stenosis. The finding of cell clusters with stem cells making the transition to cardiogenic and myocyte precursors, as well as very primitive myocytes that turn into terminally differentiated myocytes, provides a link between cardiac stem cells and myocyte differentiation. Growth and differentiation of these primitive cells was markedly enhanced in hypertrophy, consistent with activation of a restricted number of stem cells that, through symmetrical cell division, generate asynchronously differentiating progeny. These clusters strongly support the existence of cardiac stem cells that amplify and commit to the myocyte lineage in response to increased workload. Their presence is consistent with the notion that myocyte hyperplasia significantly contributes to cardiac hypertrophy and accounts for the subpopulation of cycling myocytes.
Figures


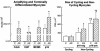
References
-
- Beltrami, A. P., Urbanek, K., Kajstura, J., Finato, N., Bussani, R., Nadal-Ginard, B., Beltrami, C. A. & Anversa, P. (2001) N. Engl. J. Med. 344, 1750-1757. - PubMed
-
- Anversa, P. & Nadal-Ginard, B. (2002) Nature 415, 240-243. - PubMed
-
- Anversa, P. & Olivetti, G. (2002) in The Heart, Handbook of Physiology, Section 2: The Cardiovascular System, eds. Page, E., Fozzard, H. & Solaro, J. (Oxford Univ. Press, New York), Vol. 1, pp. 75-144.
-
- Wagers, A. J., Christensen, J. L. & Weissman, I. L. (2002) Gene Ther. 9, 606-612. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

