Effect of codon optimization on expression levels of a functionally folded malaria vaccine candidate in prokaryotic and eukaryotic expression systems
- PMID: 12933838
- PMCID: PMC187353
- DOI: 10.1128/IAI.71.9.4961-4969.2003
Effect of codon optimization on expression levels of a functionally folded malaria vaccine candidate in prokaryotic and eukaryotic expression systems
Abstract
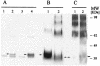
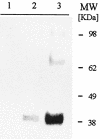
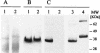
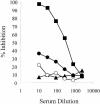
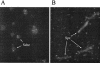
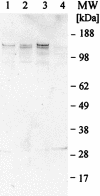
We have produced two synthetic genes that code for the F2 domain located within region II of the 175-kDa Plasmodium falciparum erythrocyte binding antigen (EBA-175) to determine the effects of codon alteration on protein expression in homologous and heterologous host systems. EBA-175 plays a key role in the process of merozoite invasion into erythrocytes through a specific receptor-ligand interaction. The F2 domain of EBA-175 is the ligand that binds to the glycophorin A receptor on human erythrocytes and is therefore a target of vaccine development efforts. We designed synthetic genes based on P. falciparum, Escherichia coli, and Pichia codon usage and expressed recombinant F2 in E. coli and Pichia pastoris. Compared to the expression of the native F2 sequence, conversion to prokaryote (E. coli)- or eukaryote (Pichia)-based codon usage dramatically improved the levels of recombinant protein expression in both E. coli and P. pastoris. The majority of the protein expressed in E. coli, however, was produced as inclusion bodies. The protein expressed in P. pastoris, on the other hand, was expressed as a secreted, soluble protein. The P. pastoris-produced protein was superior to that produced in E. coli based on its ability to bind to red blood cells. Consistent with these observations, the antibodies generated against the Pichia-produced protein prevented the binding of recombinant EBA to red blood cells. These antibodies recognize EBA-175 present on merozoites as well as in sporozoites by immunofluorescence. Our results suggest that the Pichia-based EBA-F2 vaccine construct has further potential to be developed for clinical use.
Figures

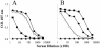
References
-
- Baird, J. K. 1995. Host age as a determinant of naturally acquired immunity to Plasmodium falciparum. Parasitol. Today 11:105-111. - PubMed
-
- Brady, C. P., R. L. Shimp, A. P. Miles, M. Whitmore, and A. W. Stowers. 2001. High-level production and purification of P30P2MSP1(19), an important vaccine antigen for malaria, expressed in the methylotropic yeast Pichia pastoris. Protein Expr. Purif. 23:468-475. - PubMed
-
- Camus, D., and T. J. Hadley. 1985. A Plasmodium falciparum antigen that binds to host erythrocytes and merozoites. Science 230:553-556. - PubMed
-
- Carrio, M. M., and A. Villaverde. 2002. Construction and deconstruction of bacterial inclusion bodies. J. Biotechnol. 96:3-12. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

