Gastric Helicobacter infection inhibits development of oral tolerance to food antigens in mice
- PMID: 12933867
- PMCID: PMC187370
- DOI: 10.1128/IAI.71.9.5219-5224.2003
Gastric Helicobacter infection inhibits development of oral tolerance to food antigens in mice
Abstract
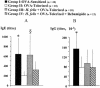
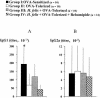
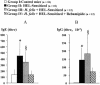
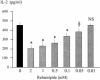
The increase in the transcellular passage of intact antigens across the digestive epithelium infected with Helicobacter pylori may interfere with the regulation of mucosal immune responses. The aim of this work was to study the capacity of Helicobacter infection to inhibit the development of oral tolerance or to promote allergic sensitization and the capacity of a gastro-protective agent, rebamipide, to interfere with these processes in mice. Oral tolerance to ovalbumin (OVA) was studied in 48 C3H/He 4-week-old mice divided into four groups: (i) OVA-sensitized mice; (ii) OVA-"tolerized" mice (that is, mice that were rendered immunologically tolerant); (iii) H. felis-infected, OVA-tolerized mice; (iv) and H. felis-infected, OVA-tolerized, rebamipide-treated mice. Oral sensitization to hen egg lysozyme (HEL) was studied in 48 mice divided into four groups: (i) controls; (ii) HEL-sensitized mice; (iii) H. felis-infected, HEL-sensitized mice; and (iv) H. felis-infected, HEL-sensitized, rebamipide-treated mice. Specific anti-OVA or anti-HEL immunoglobulin E (IgE) and IgG1/IgG2a serum titers were measured by enzyme-linked immunosorbent assay. Additionally, the capacity of rebamipide to interfere with antigen presentation and T-cell activation in vitro, as well as absorption of rebamipide across the epithelial monolayer, was tested. H. felis infection led to the inhibition of oral tolerance to OVA, but rebamipide prevented this inhibitive effect of H. felis. H. felis infection did not enhance the sensitization to HEL, but rebamipide inhibited the development of this sensitization. Moreover, rebamipide inhibited in a dose-dependent manner antigen presentation and T-cell activation in vitro and was shown to be able to cross the epithelium at a concentration capable of inducing this inhibitory effect. We conclude that H. felis can inhibit the development of oral tolerance to OVA in mice and that this inhibition is prevented by rebamipide.
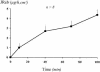
Figures
References
-
- Aihara, M., K. Imagawa, Y. Funakoshi, Y. Ohmoto, and M. Kikuchi. 1998. Effects of rebamipide on production of several cytokines by human peripheral blood mononuclear cells. Dig. Dis. Sci. 43:160S-166S. - PubMed
-
- Akamatsu, T., N. Nakamura, N. Furuya, T. Shimizu, A. Gotou, K. Kiyosawa, T. Katsuyama, T. Osumi, Y. Hirao, and G. Miyamoto. 2002. Local gastric and serum concentrations of rebamipide following oral ingestion in healthy volunteers. Dig. Dis. Sci. 47:1399-1404. - PubMed
-
- Brandtzaeg, P. 1998. Development and basic mechanisms of human gut immunity. Nutr. Rev. 56:S5-S18. - PubMed
-
- Bromander, A., J. Holmgren, and N. Lycke. 1991. Cholera toxin stimulates IL-1 production and enhances antigen presentation by macrophages in vitro. J. Immunol. 146:2908-2914. - PubMed
-
- Catto-Smith, A. G., M. K. Patrick, R. B. Scott, J. S. Davison, and D. G. Gall. 1989. Gastric response to mucosal IgE-mediated reactions. Am. J. Physiol. 257:G704-G708. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

