Ehrlichia chaffeensis and Anaplasma phagocytophilum lack genes for lipid A biosynthesis and incorporate cholesterol for their survival
- PMID: 12933880
- PMCID: PMC187327
- DOI: 10.1128/IAI.71.9.5324-5331.2003
Ehrlichia chaffeensis and Anaplasma phagocytophilum lack genes for lipid A biosynthesis and incorporate cholesterol for their survival
Abstract
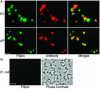
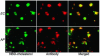
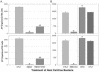
Ehrlichia chaffeensis and Anaplasma phagocytophilum are agents of human monocytic and granulocytic ehrlichioses, respectively. They are extremely sensitive to mechanical stress and are pleomorphic gram-negative bacteria. Membrane incorporation of cholesterol from the eukaryotic host is known to be essential for other fragile and pleomorphic bacteria and mycoplasmas that lack a cell wall. Thus, we tested whether cholesterol is required for E. chaffeensis and A. phagocytophilum. Using a freeze fracture technique and biochemical analysis, these bacteria were found to contain significant levels of membrane cholesterol. These bacteria lack genes for cholesterol biosynthesis or modification. However, host cell-free bacteria had the ability to take up directly exogenous cholesterol or NBD-cholesterol, a fluorescent cholesterol derivative. Treatment of the bacteria with cholesterol extraction reagent methyl-beta-cyclodextrin caused their ultrastructural changes. Furthermore, pretreatment of the bacteria with methyl-beta-cyclodextrin or NBD-cholesterol deprived these bacteria of the ability to infect leukocytes, thus killing these obligate intracellular bacteria. Analysis of E. chaffeensis and A. phagocytophilum genome sequences revealed that these bacteria lack all genes for the biosynthesis of lipid A and most genes for the biosynthesis of peptidoglycan, which confer structural strength to gram-negative bacteria. Taken together, these results suggest that human ehrlichiosis agents became cholesterol dependent due to the loss of these genes. As the first report of gram-negative bacteria incorporating cholesterol for survival, these findings offer insight into the unique nature of their parasitism and imply that cholesterol is important in the control of human ehrlichioses.
Figures


References
-
- Akira, S., K. Takeda, and T. Kaisho. 2001. Toll-like receptors: critical proteins linking innate and acquired immunity. Nat. Immunol. 2:675-680. - PubMed
-
- Andersson, S. G., A. Zomorodipour, J. O. Andersson, T. Sicheritz-Ponten, U. C. Alsmark, R. M. Podowski, A. K. Naslund, A. S. Eriksson, H. H. Winkler, and C. G. Kurland. 1998. The genome sequence of Rickettsia prowazekii and the origin of mitochondria. Nature 396:133-140. - PubMed
-
- Bakken, J. S., J. Krueth, C. Wilson-Nordskog, R. L. Tilden, K. Asanovich, and J. S. Dumler. 1996. Clinical and laboratory characteristics of human granulocytic ehrlichiosis. JAMA 275:199-205. - PubMed
-
- Banerjee, R., J. Anguita, D. Roos, and E. Fikrig. 2000. Cutting edge: infection by the agent of human granulocytic ehrlichiosis prevents the respiratory burst by down-regulating gp91phox. J. Immunol. 164:3946-3949. - PubMed
-
- Barnewall, R. E., N. Ohashi, and Y. Rikihisa. 1999. Ehrlichia chaffeensis and E. sennetsu, but not the human granulocytic ehrlichiosis agent, colocalize with transferrin receptor and up-regulate transferrin receptor mRNA by activating iron-responsive protein 1. Infect. Immun. 67:2258-2265. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

