Production of FaeG, the major subunit of K88 fimbriae, in transgenic tobacco plants and its immunogenicity in mice
- PMID: 12933900
- PMCID: PMC187361
- DOI: 10.1128/IAI.71.9.5436-5439.2003
Production of FaeG, the major subunit of K88 fimbriae, in transgenic tobacco plants and its immunogenicity in mice
Abstract
Transgenic tobacco plants stably expressing recombinant FaeG, which is the major subunit and adhesin of K88ad fimbriae, were obtained. Analysis of sera from immunized mice indicates that in mice, the immunogenicity induced by plant-derived FaeG protein is comparable to that generated with traditional approaches.
Figures
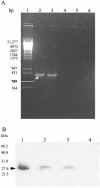
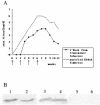
Similar articles
-
Oral immunization of mice with plant-derived fimbrial adhesin FaeG induces systemic and mucosal K88ad enterotoxigenic Escherichia coli-specific immune responses.FEMS Immunol Med Microbiol. 2006 Apr;46(3):393-9. doi: 10.1111/j.1574-695X.2005.00048.x. FEMS Immunol Med Microbiol. 2006. PMID: 16553813
-
A plasmid DNA encoding chicken interleukin-6 and Escherichia coli K88 fimbrial protein FaeG stimulates the production of anti-K88 fimbrial antibodies in chickens.Poult Sci. 2004 Dec;83(12):1973-8. doi: 10.1093/ps/83.12.1973. Poult Sci. 2004. PMID: 15615009
-
Characterization of the binding specificity of K88ac and K88ad fimbriae of enterotoxigenic Escherichia coli by constructing K88ac/K88ad chimeric FaeG major subunits.Infect Immun. 2009 Feb;77(2):699-706. doi: 10.1128/IAI.01165-08. Epub 2008 Nov 17. Infect Immun. 2009. PMID: 19015246 Free PMC article.
-
F4 fimbriae expressed by porcine enterotoxigenic Escherichia coli, an example of an eccentric fimbrial system?J Mol Microbiol Biotechnol. 2004;7(4):155-69. doi: 10.1159/000079825. J Mol Microbiol Biotechnol. 2004. PMID: 15383714 Review.
-
Development of a recombinant FimCH vaccine for urinary tract infections.Adv Exp Med Biol. 2003;539(Pt B):635-48. doi: 10.1007/978-1-4419-8889-8_41. Adv Exp Med Biol. 2003. PMID: 15176317 Review. No abstract available.
Cited by
-
Fimbrial subunit protein FaeG expressed in transgenic tobacco inhibits the binding of F4ac enterotoxigenic Escherichia coli to porcine enterocytes.Transgenic Res. 2004 Jun;13(3):295-8. doi: 10.1023/b:trag.0000034621.55404.70. Transgenic Res. 2004. PMID: 15359606
-
Immunogenicity of recombinant hepatitis B virus surface antigen fused with preS1 epitopes expressed in rice seeds.Transgenic Res. 2008 Aug;17(4):621-31. doi: 10.1007/s11248-007-9135-6. Epub 2007 Sep 20. Transgenic Res. 2008. PMID: 17882531
-
Rice MADS6 interacts with the floral homeotic genes SUPERWOMAN1, MADS3, MADS58, MADS13, and DROOPING LEAF in specifying floral organ identities and meristem fate.Plant Cell. 2011 Jul;23(7):2536-52. doi: 10.1105/tpc.111.087262. Epub 2011 Jul 22. Plant Cell. 2011. PMID: 21784949 Free PMC article.
-
Chloroplast targeting of FanC, the major antigenic subunit of Escherichia coli K99 fimbriae, in transgenic soybean.Plant Cell Rep. 2007 Jul;26(7):1011-23. doi: 10.1007/s00299-007-0322-y. Epub 2007 Mar 1. Plant Cell Rep. 2007. PMID: 17333019
-
Escherichia coli O157:H7 F9 Fimbriae Recognize Plant Xyloglucan and Elicit a Response in Arabidopsis thaliana.Int J Mol Sci. 2020 Dec 19;21(24):9720. doi: 10.3390/ijms21249720. Int J Mol Sci. 2020. PMID: 33352760 Free PMC article.
References
-
- Alexander, T. J. L. 1994. Neonatal diarrhea in pigs, p. 151-170. In C. L. Gyles (ed.), Escherichia coli in domestic animals and humans. CAB International, Wallingford, United Kingdom.
-
- Arakawa, T., J. Yu, D. K. X. Chong, J. Hough, P. C. Engen, and W. H. R. Langridge. 1998. A plant-based cholera toxin B subunit-insulin fusion protein protects against the development of autoimmune diabetes. Nat. Biotechnol. 16:934-938. - PubMed
-
- Bakker, D., C. E. M. Vader, B. Roosendaal, F. R. Mooi, B. Oudega, and F. K. de Graaf. 1991. Structure and function of periplasmic chaperone-like proteins involved in the biosynthesis of K88 and K99 fimbriae in enterotoxigenic Escherichia coli. Mol. Microbiol. 5:875-886. - PubMed
-
- Barman, N. N., and D. K. Sarma. 1999. Passive immunization of piglets against enterotoxigenic colibacillosis by vaccinating dams with K88ac pili bearing bacterins. Indian J. Exp. Biol. 37:1132-1135. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

