Shikonin, a component of chinese herbal medicine, inhibits chemokine receptor function and suppresses human immunodeficiency virus type 1
- PMID: 12936978
- PMCID: PMC182643
- DOI: 10.1128/AAC.47.9.2810-2816.2003
Shikonin, a component of chinese herbal medicine, inhibits chemokine receptor function and suppresses human immunodeficiency virus type 1
Abstract
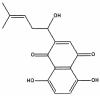
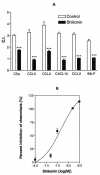
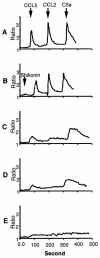
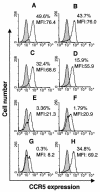
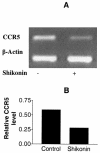
Shikonin is a major component of zicao (purple gromwell, the dried root of Lithospermum erythrorhizon), a Chinese herbal medicine with various biological activities, including inhibition of human immunodeficiency virus (HIV) type 1 (HIV-1). G protein-coupled chemokine receptors are used by HIV-1 as coreceptors to enter the host cells. In this study, we assessed the effects of shikonin on chemokine receptor function and HIV-1 replication. The results showed that, at nanomolar concentrations, shikonin inhibited monocyte chemotaxis and calcium flux in response to a variety of CC chemokines (CCL2 [monocyte chemoattractant protein 1], CCL3 [macrophage inflammatory protein 1alpha], and CCL5 [regulated upon activation, normal T-cell expressed and secreted protein]), the CXC chemokine (CXCL12 [stromal cell-derived factor 1alpha]), and classic chemoattractants (formylmethionyl-leucine-phenylalanine and complement fraction C5a). Shikonin down-regulated surface expression of CCR5, a primary HIV-1 coreceptor, on macrophages to a greater degree than the other receptors (CCR1, CCR2, CXCR4, and the formyl peptide receptor) did. CCR5 mRNA expression was also down-regulated by the compound. Additionally, shikonin inhibited the replication of a multidrug-resistant strain and pediatric clinical isolates of HIV in human peripheral blood mononuclear cells, with 50% inhibitory concentrations (IC(50)s) ranging from 96 to 366 nM. Shikonin also effectively inhibited the replication of the HIV Ba-L isolate in monocytes/macrophages, with an IC(50) of 470 nM. Our results suggest that the anti-HIV and anti-inflammatory activities of shikonin may be related to its interference with chemokine receptor expression and function. Therefore, shikonin, as a naturally occurring, low-molecular-weight pan-chemokine receptor inhibitor, constitutes a basis for the development of novel anti-HIV therapeutic agents.
Figures



Similar articles
-
Shikonin, a component of antiinflammatory Chinese herbal medicine, selectively blocks chemokine binding to CC chemokine receptor-1.Int Immunopharmacol. 2001 Feb;1(2):229-36. doi: 10.1016/s1567-5769(00)00033-3. Int Immunopharmacol. 2001. PMID: 11360924
-
Inhibition of human immunodeficiency virus replication by a dual CCR5/CXCR4 antagonist.J Virol. 2004 Dec;78(23):12996-3006. doi: 10.1128/JVI.78.23.12996-13006.2004. J Virol. 2004. PMID: 15542651 Free PMC article.
-
CXCR4 and CCR5 expression by H9 T-cells is downregulated by a peptide-nucleic acid immunomodulator.Immunol Lett. 2000 Nov 1;74(3):189-95. doi: 10.1016/s0165-2478(00)00258-3. Immunol Lett. 2000. PMID: 11064099
-
Small molecule inhibitor of HIV-1 cell fusion blocks chemokine receptor-mediated function.J Leukoc Biol. 1998 Jul;64(1):6-13. doi: 10.1002/jlb.64.1.6. J Leukoc Biol. 1998. PMID: 9665268 Review.
-
Chemokine receptor-directed agents as novel anti-HIV-1 therapies.Curr Top Med Chem. 2004;4(10):1017-33. doi: 10.2174/1568026043388277. Curr Top Med Chem. 2004. PMID: 15193136 Review.
Cited by
-
Shikonin Inhibites Migration and Invasion of Thyroid Cancer Cells by Downregulating DNMT1.Med Sci Monit. 2018 Feb 1;24:661-670. doi: 10.12659/msm.908381. Med Sci Monit. 2018. PMID: 29389913 Free PMC article.
-
Anxiolytic and Hypnotic Effects of Aqueous and Ethanolic Extracts of Aerial Parts of Echium italicum L. in Mice.Jundishapur J Nat Pharm Prod. 2012 Spring;7(2):71-9. Epub 2012 May 28. Jundishapur J Nat Pharm Prod. 2012. PMID: 24624158 Free PMC article.
-
HIV entry inhibitors and their potential in HIV therapy.Med Res Rev. 2009 Mar;29(2):369-93. doi: 10.1002/med.20138. Med Res Rev. 2009. PMID: 18720513 Free PMC article. Review.
-
Astragalus polysaccharides inhibit avian infectious bronchitis virus infection by regulating viral replication.Microb Pathog. 2018 Jan;114:124-128. doi: 10.1016/j.micpath.2017.11.026. Epub 2017 Nov 21. Microb Pathog. 2018. PMID: 29170045 Free PMC article.
-
YTHDC1 Regulates the Migration, Invasion, Proliferation, and Apoptosis of Rheumatoid Fibroblast-Like Synoviocytes.Front Immunol. 2024 Oct 29;15:1440398. doi: 10.3389/fimmu.2024.1440398. eCollection 2024. Front Immunol. 2024. PMID: 39534605 Free PMC article.
References
-
- Baba, M., O. Nishimura, N. Kanzaki, M. Okamoto, H. Sawada, Y. Iizawa, M. Shiraishi, Y. Aramaki, K. Okonogi, Y. Ogawa, K. Meguro, and M. Fujino. 1999. A small-molecule, nonpeptide CCR5 antagonist with highly potent and selective anti-HIV-1 activity. Proc. Natl. Acad. Sci. USA 96:5698-5703. - PMC - PubMed
-
- Buckheit, R. W., Jr., V. Fliakas-Boltz, W. D. Decker, J. L. Roberson, C. A. Pyle, E. L. White, B. J. Bowdon, J. B. McMahon, M. R. Boyd, J. P. Bader, et al. 1994. Biological and biochemical anti-HIV activity of the benzothiadiazine class of nonnucleoside reverse transcriptase inhibitors. Antivir. Res. 25:43-56. - PubMed
-
- Buckheit, R. W., Jr., T. L. Kinjerski, V. Fliakas-Boltz, J. D. Russell, T. L. Stup, L. A. Pallansch, W. G. Brouwer, D. C. Dao, W. A. Harrison, R. J. Schultz, et al. 1995. Structure-activity and cross-resistance evaluations of a series of human immunodeficiency virus type-1-specific compounds related to oxathiin carboxanilide. Antimicrob. Agents Chemother. 39:2718-2727. - PMC - PubMed
-
- Buckheit, R. W., Jr., J. L. Roberson, C. Lackman-Smith, J. R. Wyatt, T. A. Vickers, and D. J. Ecker. 1994. Potent and specific inhibition of HIV envelope-mediated cell fusion and virus binding by G quartet-forming oligonucleotide (ISIS 5320). AIDS Res. Hum. Retrovir. 10:1497-1506. - PubMed
-
- Carpenter, C. C., D. A. Cooper, M. A. Fischl, J. M. Gatell, B. G. Gazzard, S. M. Hammer, M. S. Hirsch, D. M. Jacobsen, D. A. Katzenstein, J. S. Montaner, D. D. Richman, M. S. Saag, M. Schechter, R. T. Schooley, M. A. Thompson, S. Vella, P. G. Yeni, and P. A. Volberding. 2000. Antiretroviral therapy in adults: updated recommendations of the International AIDS Society—USA Panel. JAMA 283:381-390. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

